short course on clinical investigation for medical devices and iso 14155
Published 2 years ago • 7.3K plays • Length 19:26Download video MP4
Download video MP3
Similar videos
-
 4:00
4:00
the responsibilities of a clinical investigation sponsor
-
 0:41
0:41
bloopers: recording introduction to clinical investigation for medical devices and iso 14155
-
 8:27
8:27
clinical investigation and clinical evaluation of medical devices
-
 3:54
3:54
the value of the iso 14155 standard for clinical investigations
-
 3:47
3:47
clinical research for your medical devices
-
 14:55
14:55
short course on the medical device regulation (eu) 2017/745
-
 4:06
4:06
how to do a clinical investigation in less than 12 months
-
 6:44
6:44
what are the major changes in iso 14155 2020?
-
 37:48
37:48
how to perform a clinical investigation for your medical devices?
-
 2:53
2:53
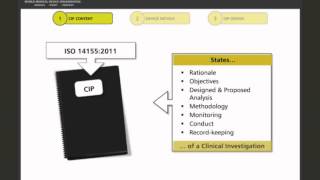
format and structure of a medical device clinical investigation plan/protocol
-
 17:07
17:07
risk management for medical devices and iso 14971 - online introductory course
-
 3:33
3:33
citi program webinar demo - understanding iso 14155:2020 revisions
-
 41:00
41:00
clinical investigation of medical devices, regulation of investigational medical devices
-
 12:49
12:49
process validation for medical devices - short course
-
 2:33
2:33
gcp for investigators: initiation of a medical device clinical investigation
-
 2:32
2:32
introduction to medical device and clinical investigation planning
-
 6:39
6:39
what is good clinical practice (gcp)?