short course on samd (software as a medical device), iec 62304 and iec 82304-1
Published 1 year ago • 9.9K plays • Length 28:40Download video MP4
Download video MP3
Similar videos
-
 23:06
23:06
medical device software development short course
-
 2:17
2:17
samd (software as medical device) 101: 3 types of validations
-
 5:43
5:43
what is software as a medical device (samd) in clinical research?
-
 12:25
12:25
medical device software risk management and iec 62304 terminology
-
 12:24
12:24
introduction to different classifications rules for medical device software
-
 12:34
12:34
documenting compliance with iec 62304 in medical device software development
-
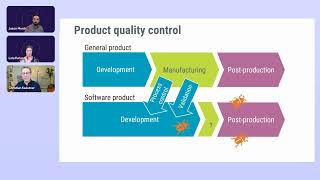 58:53
58:53
software as medical device (samd): ask the expert
-
 16:45
16:45
iacm overview demo
-
 32:54
32:54
179 samd vivek thakkar
-
 9:31
9:31
system, item and units in medical device software
-
 10:44
10:44
managing and documenting soup and ots in medical device software
-
 14:56
14:56
risk control measures in medical device software
-
 14:55
14:55
short course on the medical device regulation (eu) 2017/745
-
 51:51
51:51
webinar: risk management in software as a medical device (samd) development
-
 33:33
33:33
optimize your quality system for software as a medical device (samd)
-
 1:11:09
1:11:09
how to do software as medical device development correctly?
-
 12:44
12:44
safety for electrical medical devices - short course
-
 27:12
27:12
what is a soup for your software development
-
 26:07
26:07
software as a medical device