show that the minimum cation-to-anion radius ratio for a coordination number of 6 is 0.4142
Published 2 months ago • 338 plays • Length 5:15Download video MP4
Download video MP3
Similar videos
-
 7:10
7:10
calculation of cation-anion radius ratio based on coordination number
-
 5:38
5:38
cation - anion radius ratio for coordination number 3
-
 6:42
6:42
calculating cation-anion radius ratios for coordination number 8
-
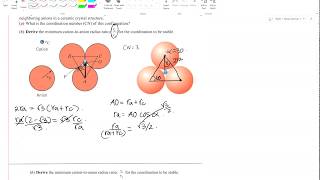 7:30
7:30
deriving cation-anion ratio for coordination numbers 3,6 and 8
-
 11:23
11:23
calculating cation-anion radius ratios for tetrahedral coordination number 4
-
 8:55
8:55
cation to anion radius ratio for tetrahedral interstitial site
-
 5:21
5:21
calculating coordination number from ion size and predicting crystal structure example problem
-
 22:44
22:44
mineralogy: lecture 18, coordination number
-
 9:27
9:27
18 the octahedral site
-
 27:56
27:56
ratio test
-
 6:59
6:59
radius ratio rule
-
 0:59
0:59
jl chemistry. solid state coordination number.radius
-
 15:15
15:15
1 crystal structure: 1.1ligancy 6 & 8 #ligancysix #ligancyeight #ligancy #criticalradiusratio
-
 3:19
3:19
bonding 6: coordination number determination
-
 3:21
3:21
if the radius ratio of cation to anion is in the range of 0.225 - 0.414 , then the
-
 3:09
3:09
solids: radius ratio rule
-
 9:03
9:03
pauling's rule 1. coordination principle (radius ratios)
-
 4:51
4:51
the cation/anion radius ratio for a triangular arrangement of anions in which the cation is in c...
-
 3:23
3:23
coordination compounds: coordination number: lecture 4
-
 12:08
12:08
coordination number of simple cubic, fcc, bcc and hcp lattice
-
 11:15
11:15
radius ratio – tetrahedral void, problems