st101 lecture 4: development and validation of stability indicating methods
Published 9 years ago • 2K plays • Length 6:35Download video MP4
Download video MP3
Similar videos
-
 16:57
16:57
stability indicating method development and validation of the trandolapril in human plasma
-
 2:53
2:53
st101 lecture 14: stability to support post approval changes
-
 1:31
1:31
st101 lecture 11: evaluation of stability data
-
 1:19
1:19
st101 lecture 9: stability program for biologics
-
 54:33
54:33
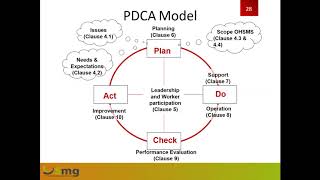
iso 45001 lead auditor training/ iso 45001-2018/ #osh #iso45001 #iso #leadauditor
-
 38:32
38:32
iso 45001:2018 occupational health and safety management system
-
 55:16
55:16
practical aspects of hplc method development
-
 1:43
1:43
st101 lecture 1: introduction to stability
-
 3:14
3:14
st101 lecture 5: monitoring organic impurities during stability program
-
 3:04
3:04
st101 lecture 12: using stability data to support specification setting
-
 2:06
2:06
st101 lecture 3: qbd approach for stability
-
 2:16
2:16
st101 lecture 6: photostability testing and q1b
-
 2:00
2:00
st101 lecture 10: bracketing and matrixing of stability testing
-
 1:59
1:59
st101 lecture 8: stability expectations for generic drugs (andas)
-
 1:45
1:45
st101 lecture 2: regulations on stability program global stability requirements
-
 5:03
5:03
stability indicating methods
-
 1:04
1:04
st101 lecture 7: stability of parenterals
-
 1:11:51
1:11:51
110122 criticality of stability indicating hptlc method development
-
 2:11
2:11
study on development and validation of stability indicating rp hplc method for guaifenesin
-
 7:36
7:36
validated stability indicating rp-hplc and hptlc methods for the determination of zanamivir in bulk