identify the spectator ions in the following reaction: ca2 (aq) 2no3-(aq) 2na (aq) co3^2-(aq)…
Published 3 weeks ago • 1 view plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 8:15
8:15
12.6 | consider the following reaction in aqueous solution: 5br−(aq) bro3−(aq) 6h (aq) →
-
 0:33
0:33
you have the following reaction: 3au (s) 8h (aq) 2no3 (aq) - 3au2 (aq) 2no (g) 4h2o (liq…
-
 0:33
0:33
using these molar enthalpy values: (1) mgo(s) 2 hcl(aq) → mgcl2(aq) h2o(aq) î”h1 = -134 kj/mo…
-
 0:33
0:33
the reaction between hcl(aq) and nahco3(aq) produces all of the following compounds except ________…
-
 0:33
0:33
consider the reaction below. al(s) cu2 (aq) ? al3 (aq) cu(s) choose the reaction that is balanc…
-
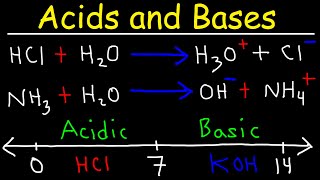 58:42
58:42
acids and bases - basic introduction - chemistry
-
 29:17
29:17
kinetic monte carlo simulations of atomic layer deposition
-
 36:58
36:58
500 years of not teaching the cubic formula. what is it they think you can't handle?
-
 2:35
2:35
ch_3-ch_2-br koh_((aq)) to \'a\' यहाँ \'a\' है | 12 | मॉडल पेपर 2021सेट 02 | chemistry | jhar...
-
 10:31
10:31
4.39a | balance: sn2 (aq) cu2 (aq) → sn4 (aq) cu (aq)
-
 0:33
0:33
calculate the equilibrium constant for the reaction, fe(s) ni2 (aq) → fe2 (aq) ni(s). ( ) 2.8…
-
 0:33
0:33
25 fe(s) 2 hci(aq) - fecl2(aq) h2(g) when a student adds 30.0 ml of 1.00 m hci to 0.56 g of po…
-
 0:11
0:11
the density of two liquids (a and b) is given as 1000 kg/m3 and 600 kg/m3, respectively. the two liq
-
 0:33
0:33
the following reactions 2k(s) br2(2kbr(s)), agno3(aq) nacl(aq) agcl(s) nano3(aq), hcl(aq) k…
-
 13:56
13:56
form 5 (chapter 2.3.1(ii))
-
 4:59
4:59
17.21b | calculate the standard cell potential for 2ag (aq) cu(s) → cu2 (aq) 2ag(s)
-
 8:04
8:04
a 2b 3c⇌ab2c3 reaction of \(6 g\) of \(a , 6 \times 10^{23}\) atoms of b & 0.036 mole ....
-
 0:33
0:33
the average translational kinetic energy of an atom or molecule is about _ke = 3/…
-
 1:27
1:27
balancing the equation al cl2 = alcl3
-
 3:23
3:23
the reaction, 2a(g) b(g)⇌3c(g) d(g) is begun with the concentrations of a and b both at an initial
-
 15:43
15:43
4.11b | write the net ionic equation: pb(no3)2(aq) h2so4(aq) → pbso4(s) 2hno3(aq)