the equilibrium constant for the reaction ni2 (aq) 6 nh3(aq) ? ni(nh3)6 2 (aq) is kf = 5.6 × 108 …
Published 4 days ago • No plays • Length 0:33
Download video MP4
Download video MP3
Similar videos
-
 0:33
0:33
the equilibrium constant for the reaction ni (s) hg2cl2 (s) → 2hg (l) 2cl^-(aq) ni2 (aq) is…
-
 0:33
0:33
calculate the equilibrium constant for the reaction, fe(s) ni2 (aq) → fe2 (aq) ni(s). ( ) 2.8…
-
 0:33
0:33
for the chemical equilibrium a 2b → 2c, the value of the equilibrium constant, k, is 1.55 ã— 10…
-
 0:33
0:33
the equilibrium constant kc for the reaction i2(g) ⇆ 2i(g) is 3.85 ã— 10^(-5) at 725 â°c. calcula…
-
 0:33
0:33
a reaction vessel contains nh3, n2, and h2 at equilibrium at a certain temperature. the equilibrium…
-
 0:33
0:33
calculate the equilibrium constant at 25â°c for the reaction: 3 tl(aq) → tl3 (aq) 2 tl(s) e = -…
-
 3:31
3:31
predict equilibrium constant from two other reactions
-
 6:41
6:41
determining equilibrium pressures from kp 007
-
 13:58
13:58
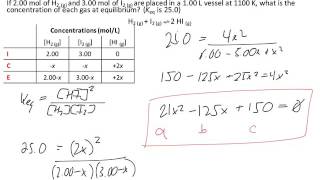
quadratic equation ice table equilibrium calculations
-
 0:33
0:33
the change in enthalpy (î”horxn) for a reaction is -29.7 kj/mol. the equilibrium constant for the r…
-
 0:33
0:33
3. in the reaction below, nh3(g) h2o (l) ? nh2- h3o , the equilibrium constant is 10-34. is thi…
-
 0:33
0:33
ge(g) 2 cl2(g) gecl2(g) 7. the value of the equilibrium constant for the reaction represented abo…
-
 0:33
0:33
for the following reaction, kp = 137 at 3.50 x 10^2 k. what is the value of kc? 2c4h10(g) - 8co2(g…
-
 0:33
0:33
calculate the value of the equilibrium constant, k, for a reaction with î”hâ° = -95 kj/mol and î”sâ…
-
 0:33
0:33
calculate the equilibrium constant, k, for the reaction shown at 25 â°c. fe3 (aq) b(s) 6h2o(l) …
-
 0:33
0:33
the equilibrium constant, k, for the following reaction is 1.80×10-4 at 298 k. nh4hs(s) nh3(g) h2…
-
 0:33
0:33
equilibrium constant for a reaction that has a value of what is the = -41.8 kj at 100â°c? (r = 0.08…
-
 0:33
0:33
fe3 (aq) scn-(aq) – fescn2 (aq) 1. write the equilibrium constant expression for the reaction if…
-
 0:33
0:33
the value of the equilibrium constant kp for the reaction below is 0.657 at 900â°c. h2(g) co2(g) …̊
-
 0:33
0:33
1s22522p63s23ps at 300 = 'c. the 16. the 'equilibrium constant for the production of ammonia gas wa…
-
 0:33
0:33
if a reducing agent m reacts with an oxidizing agent n to give m and n, and the equilibrium const…
-
 0:33
0:33
it is observed that after two half-lives have passed, one-third of the original material is left. w…
Clip.africa.com - Privacy-policy
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33
 3:31
3:31
 6:41
6:41
 13:58
13:58
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33