equilibrium constant, `k_(c)` for the reaction, `n_(2(g)) 3h_(2(g))harr2nh_(3(g))`, at `500 k`
Published 3 years ago • 295 plays • Length 2:18Download video MP4
Download video MP3
Similar videos
-
 53:22
53:22
chemical equilibrium constant k - ice tables - kp and kc
-
 10:51
10:51
how to calculate kp from kc - chemical equilibrium
-
 7:06
7:06
equilibrium constant, kc for the reaction, n2 (g) 3h2 (g) ⇌ 2nh3 (g); at 500k is 0.061......
-
 2:23
2:23
n2(g) 3h2(g) ⇌ 2nh3(g); δh° = -92 kj
-
 1:39
1:39
the equilibrium constant for the reaction, n2 3h2 ⇌ 2nh3 is kc
-
 7:31
7:31
how to write equilibrium constant expressions | kc keq kp | basics chemical equilibrium
-
 34:23
34:23
equilibrium constant kp | a level chemistry | explained
-
 6:15
6:15
the equilibrium constant
-
 2:10
2:10
the equilibrium constants for the following reactions n_(2) (g) 3h_(2)(g) harr 2nh_(3) (g) n_(...
-
 30:20
30:20
13.82 | the equilibrium constant (kc) for this reaction is 1.60 at 990 °c: h2(g) co2(g) ⇌ h2o(g)
-
 5:48
5:48
the equilibrium constant kc, for 2so3(g) ↔ 2so2(g) o2(g) is 2.9x10- 4 at 1150 k. calculate kp
-
 8:24
8:24
13.19 | the following reaction has kp = 4.50 × 10^−5 at 720 k. n2(g) 3h2(g) ⇌ 2nh3(g)
-
 10:27
10:27
the equilibrium constant kc
-
 13:26
13:26
3 equilibrium constant kc&kp
-
 6:34
6:34
how to calculate the equilibrium constant (kc) for a reaction
-
 12:30
12:30
kp and kc
-
 36:47
36:47
equilibrium constant, kc | a level chemistry
-
 3:02
3:02
practice problem: calculating equilibrium concentrations
-
 15:53
15:53
equilibrium constant: kc | a-level chemistry | ocr, aqa, edexcel
-
 6:40
6:40
13.25a | convert the values of kc to values of kp: n2(g) 3h2(g) ⇌ 2nh3(g) kc = 0.50 at 400 °c
-
 2:49
2:49
college chemistry: write the kc and kp expression
-
 1:40
1:40
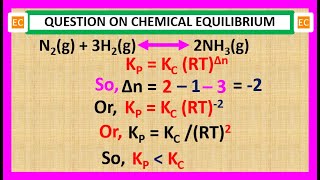
oqv no – 36 relation between kp and kc for the reaction n2 3h2 = 2nh3.