the following reaction is found to obey a second order rate law where the partial a 2b kz orders o…
Published 3 months ago • No plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 0:33
0:33
the reaction for the decomposition of no2 is found to follow second-order kinetics. at a certain te…
-
 0:33
0:33
the rate constant of a specific second-order reaction is 0.0143/m.s. if the initial concentration o…
-
 0:33
0:33
which one of the following statements does not describe the equilibrium state? a) equilibrium is dy…
-
 5:17
5:17
find the order of the reaction example
-
 0:33
0:33
the following kinetic data ( v_0 is the initial rate) were obtained for the reaction 2 ic…
-
 25:16
25:16
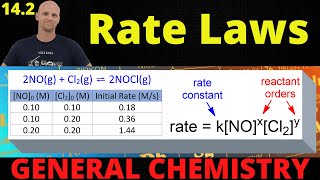
14.2 rate laws | general chemistry
-
 5:45
5:45
chemistry - chemical kinetics (1 of 30) definition
-
 5:45
5:45
order of reaction
-
 0:33
0:33
how many moles of c are formed upon complete reaction of 2 mol of b according to the generic chemic…
-
 0:33
0:33
the following reaction is first order: c2h6 ? 2 ch3. if the rate constant is equal to 5.5 × 10-4 s-…
-
 0:33
0:33
2no(g) o2(g) → 2no2(g) for the above reaction, the following data were collected for the rate o…
-
![consider the following hypothetical reaction and the established rate law: a bx rate k[a] [b] s…](https://i.ytimg.com/vi/wnUY22TwHWg/mqdefault.jpg) 0:33
0:33
consider the following hypothetical reaction and the established rate law: a bx rate k[a] [b] s…
-
 0:33
0:33
consider a hypothetical biochemical pathway involving the following reactions, which occur in seque…
-
 0:33
0:33
the data shown below were collected for the following first-order reaction: part a n2o(g) - n2(g) …
-
 3:30
3:30
chemistry - chemical kinetics (8 of 30) reaction order - how are they determined?
-
 0:33
0:33
draw the major organic product(s) of the following reactions including stereochemistry when it is a…
-
 0:33
0:33
1. the following equation is used to calculate specific heat: q = cmî”t where î”t represents the ch…
-
 4:09
4:09
determine the half-life of a second order reaction
-
 0:33
0:33
using the method of initial rates, determine the order with respect to the hydroxide ion. do this b…
-
 0:33
0:33
during a chemical reaction, the function y = f(t) models the amount of substance present in grams o…
-
 0:33
0:33
consider the following reaction: p4(g) 5 o2(g) → p4o10(s) the enthalpy change at 298 k for this…
-
 0:33
0:33
draw the structure resulting from the reaction of osmium tetroxide (oso4) and hydrogen peroxide (h2…