21.54 | the mass of a hydrogen atom (h-1) is 1.007825 amu; that of a tritium atom (h-3) is 3.01605
Published 9 months ago • 376 plays • Length 15:13
Download video MP4
Download video MP3
Similar videos
-
 7:15
7:15
the atomic masses of the hydrogen isotopes are hydrogen `m_1h^1=1.007825`
-
 0:37
0:37
hydrogen’s 3 isotopes: protium, deuterium and tritium ⚛️
-
 2:09
2:09
how to find the mass of one atom of hydrogen (h)
-
 3:35
3:35
isotopes of hydrogen (protium, deuterium, and tritium)
-
 7:37
7:37
tritium is the `.^(3)h` isotope of hydrogen. its atomic mass is 3.016amu: the atomic mass of `.^
-
 0:33
0:33
what is the fraction of the hydrogen atom's mass (â¹â¹h) that is in the nucleus? the mass of a prot…
-
 1:41
1:41
three isotopes of hydrogen, deuterium, tritium
-
 7:59
7:59
physics - ch 66.5 quantum mechanics: the hydrogen atom (41 of 78) what is the reduced mass?
-
 1:47
1:47
physics - ch 66.5 quantum mechanics: the hydrogen atom (43 of 78) the reduced mass correction
-
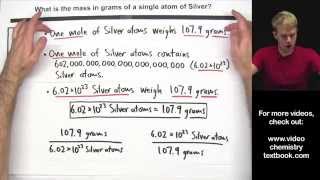 7:16
7:16
calculate the mass of a single atom or molecule
-
 12:24
12:24
radioactivity (5 of 16) nuclear fusion, an explanation
-
 5:09
5:09
physics - ch 66.5 quantum mechanics: the hydrogen atom (56 of 78) solution of the theta fct: c3 p1
-
 3:45
3:45
physics - ch 66.5 quantum mechanics: the hydrogen atom (57 of 78) solution of the theta fct: c3 p2
-
 1:59
1:59
atomic number for protium, deuterium, and tritium
-
 7:28
7:28
ram iii: atomic mass units
-
 3:44
3:44
physics - ch 66.5 quantum mechanics: the hydrogen atom (55 of 78) solution of the theta fct: c2 p2
-
 6:16
6:16
physics - ch 66.5 quantum mechanics: the hydrogen atom (58 of 78) solution of the theta fct: c4 p1
-
 4:25
4:25
physics - ch 66.5 quantum mechanics: the hydrogen atom (52 of 78) solution of the theta fct
-
 2:07
2:07
deuterium - heavy hydrogen - isotopes of hydrogen (protium, deuterium, and tritium)
Clip.africa.com - Privacy-policy
 7:15
7:15
 0:37
0:37
 2:09
2:09
 3:35
3:35
 7:37
7:37
 0:33
0:33
 1:41
1:41
 7:59
7:59
 1:47
1:47
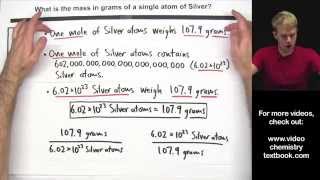 7:16
7:16
 12:24
12:24
 5:09
5:09
 3:45
3:45
 1:59
1:59
 7:28
7:28
 3:44
3:44
 6:16
6:16
 4:25
4:25
 2:07
2:07