the rate of the reaction, 2no cl2→2nocl is given by the rate equation, rate =k[no2 [ cl2 ].
Published 2 months ago • 74 plays • Length 0:51Download video MP4
Download video MP3
Similar videos
-
 7:36
7:36
determine rate equation and rate constant of the reaction 2no cl2=2nocl using table of initial rates
-
 0:57
0:57
find the value of kc if kp for the given reaction is 1.8×10−2 at 500k 2nocl⇌2no cl2.
-
 0:47
0:47
determine the rate equation of the reaction 2no(g) cl2(g) = 2nocl(g)#chemistry #classroom #college
-
 0:33
0:33
the gas phase reaction 2no cl2 → 2nocl is believed to be truly termolecular. the temperature de…
-
 10:56
10:56
calculating rates ii | general chemistry ii | 5.4
-
 4:22
4:22
the rate of reaction: `2no(g) cl_(2) (g) rarr 2nocl (g)`, becomes doubled when the concentration
-
 8:42
8:42
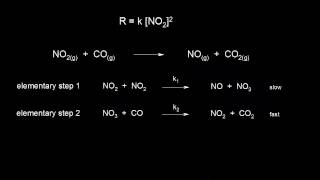
reaction mechanism and rate law | kinetics | ap chemistry | khan academy
-
 18:44
18:44
reaction mechanisms, rate laws, reaction profiles, and sn1 vs. sn2 reactions
-
 34:53
34:53
chemical kinetics - initial rates method
-
 23:43
23:43
12.25 | nitrogen monoxide reacts with chlorine according to the equation: 2no(g) cl2(g) → 2nocl(g)
-
 1:51
1:51
for the reaction `2nocl(g) harr 2no(g) cl_(2)(g)`, the equilibrium constant is `2.8xx10^(-5)` at...
-
 2:42
2:42
r2.2.11 the rate constant k (hl)
-
 9:10
9:10
kinetics: initial rates and integrated rate laws
-
 3:18
3:18
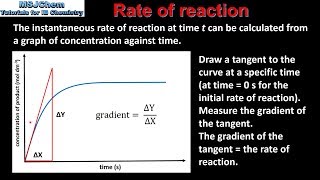
r2.2.1 - how do we calculate the rate of reaction from a time-concentration graph?
-
 3:58
3:58
16.2.1 / 16.2.2 rate determining step
-
 0:33
0:33
the rate law for the reaction 2 h_2(g) 2 no(g) ⟶n_2(g…
-
 6:04
6:04
16.1.2 deduce the rate expression for a reaction from experimental data.
-
 9:20
9:20
more than one reactant | general chemistry ii | 6.6
-
 2:37
2:37
r2.2.1 rate of reaction
-
 5:35
5:35
chemistry - chemical kinetics (2 of 30) reaction rate- definition