the reaction, n2 3h2 → 2nh3 is used to produce ammonia.
Published 6 years ago • 18K plays • Length 1:23Download video MP4
Download video MP3
Similar videos
-
 1:16
1:16
consider the reaction 2nh3(g) → n2(g) 3h2(g)
-
 0:33
0:33
part a- ammonia can be produced via the chemical reaction n2(g) 3h2(g) ⇜ 2nh3(g) during the pro…
-
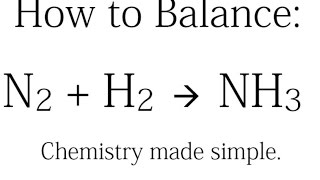 1:00
1:00
how to balance: n2 h2 = nh3 (synthesis of ammonia)
-
 1:39
1:39
the equilibrium constant for the reaction, n2 3h2 ⇌ 2nh3 is kc
-
![consider the following reaction n2 3h2— 2nh3 the rate of reaction in terms of n2 is -d[n2]/dt = 0.02](https://i.ytimg.com/vi/smQ8fuXlxtw/mqdefault.jpg) 3:26
3:26
consider the following reaction n2 3h2— 2nh3 the rate of reaction in terms of n2 is -d[n2]/dt = 0.02
-
 17:40
17:40
tricks to solve thermochemistry problems easily | enthalpy of formation combustion
-
 20:12
20:12
stoichiometry - limiting & excess reactant, theoretical & percent yield - chemistry
-
 3:37
3:37
form 4 (chapter 6.9.3)
-
 0:53
0:53
unit of kp for the reaction n2(g) 3h2(g) ↔ 2nh3(g). #chemistry
-
 0:33
0:33
in the haber process, ammonia is synthesized from nitrogen and hydrogen: n2 (g) 3h2 (g) → 2nh3 …
-
 2:10
2:10
for the reaction n2 3h2 gives 2nh3, kc depends on
-
 0:33
0:33
in the haber process, hydrogen (h2) and nitrogen (n2) react to produce ammonia (nh3): h2 n2 —-…
-
 5:55
5:55
ammonia gas decomposes according to the equation: 2nh3(g) → n2(g) 3h2(g). if 17.5 l of nitrogen is
-
 6:38
6:38
the %yield of ammonia as a function of time in the reaction : n2(g) 3h2(g)⇌2nh3(g), #jee2015adv.
-
 0:49
0:49
homogeneous reaction n2(g) 3h2(g) → 2nh3(g). #chemistry
-
 2:23
2:23
n2(g) 3h2(g) ⇌ 2nh3(g); δh° = -92 kj
-
 1:08
1:08
n2 3h2 2nh3. in this reaction equivalent weight of n2 is
-
 5:10
5:10
16.38a | would ammonia be a suitable candidate for fuels: 2nh3(g) → n2(g) 3h2(g)
-
 12:30
12:30
kp and kc
-
 4:53
4:53
chemical equilibrium of ammonia production and use of le chatelier's principle
-
 0:33
0:33
nitrogen (n) and hydrogen (h) react with each other to produce ammonia (nh3). this reaction is show…
-
 2:38
2:38
which one of the following equations is correct for the reaction