the solubility product constant of calcium chlorate, ca(clo3)) of water is 7.1 * 10 ^ - 7 at 25 . h…
Published 2 days ago • No plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 8:36
8:36
solubility product constant (ksp)
-
 20:25
20:25
14.04 the solubility product constant
-
 8:20
8:20
ksp and molar solubility calculations video
-
 4:21
4:21
8.3 ionic product constant of water kw (sl)
-
 5:26
5:26
the solubility product
-
 4:40
4:40
solubility product constant with example
-
 1:52:54
1:52:54
masquelier, barker | crystal chemistry of phosphate-containing & commercialization of sodium-ion
-
 2:09:07
2:09:07
the soil carbon sponge, climate solutions and healthy water cycles with walter jehne
-
 4:53
4:53
lab 12 ksp determination
-
 0:33
0:33
from the solubility product constant of zinc ferrocyanide, zn2fe(cn)6, calculate the concentration …
-
 0:31
0:31
what is a solubility product ksp @diarasacademy
-
 1:10
1:10
solubility product (ksp) sample problem: chapter 17 – part 11
-
 3:03
3:03
practice problem: solubility product constant calculations
-
 5:03
5:03
r3.1.5 ionic product constant of water, kw
-
 1:31
1:31
understanding the solubility product constant
-
 0:24
0:24
solubility 1-4
-
 2:00
2:00
ksp - solubility product constant molar solubility: example 1
-
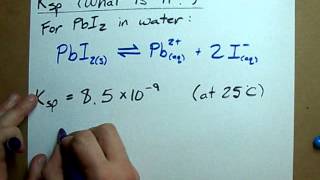 3:40
3:40
what is ksp? (solubility product constant)
-
 10:04
10:04
solubility product and predicting precipitation // hsc chemistry
-
 12:38
12:38
equilibrium: the solubility product
-
 2:17
2:17
solubility product - calculating the concetration