the standard emf for the cell using the overall cell reaction below is 2.20 v: 2al (s) 3i 2 (s) …
Published 3 days ago • No plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 10:56
10:56
cell potential problems - electrochemistry
-
 0:33
0:33
overall reaction: pb(s) pbo2(s) 2 h (aq) 2 hso4-(aq) → 2 pbso4(s) 2 h2o(l) cathode half-c…
-
 0:33
0:33
consider the reaction corresponding to a voltaic cell and its standard cell potential. z n ( s ) …
-
![calculate the cell potential for the reaction as written at 25.00 â°c, given that [zn2 ]=0.791 m an…](https://i.ytimg.com/vi/ran9tlV9qK8/mqdefault.jpg) 0:33
0:33
calculate the cell potential for the reaction as written at 25.00 â°c, given that [zn2 ]=0.791 m an…
-
 5:30
5:30
17.22a | calculate the standard cell potential for mn(s) ni2 (aq) → mn2 (aq) ni(s)
-
 3:35
3:35
trick to identify anode and cathode in a cell reaction
-
 13:15
13:15
determining od600 (cell density)
-
 16:37
16:37
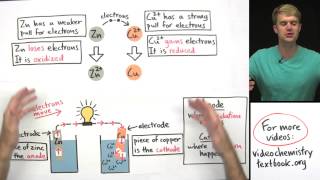
introduction to electrochemistry
-
 0:33
0:33
urgent plz find the cell potential for each of the below reactions: a. cu(s) znso4(aq) – zn(s) …
-
 0:33
0:33
calculate the standard free energy change for the following reactions using the standard cell poten…
-
 14:22
14:22
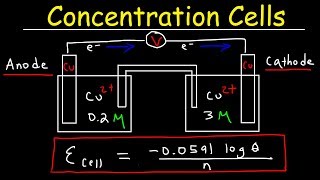
concentration cells & cell potential calculations - electrochemistry
-
 5:28
5:28
how to calculate standard cell potential and voltage using e cell = e cathode - e anode examples
-
 5:27
5:27
r3.2.13 calculating cell potential (hl)
-
 0:33
0:33
the reduction potential for all other half-reactions is measured relative to the half-reaction 2h (…
-
 4:54
4:54
how to find the cell potential (ecell) under standard conditions
-
 0:33
0:33
hydrogen sulfide is composed of two elements
-
 9:43
9:43
calculating the equilibrium constant from the standard cell potential | khan academy
-
 0:33
0:33
5.0g of nitrogen is reacted with 5.0 g of hydrogen to produce ammonia according to the chemical equ…
-
![1.) balance this skeleton reaction: mn2 (aq) co3 (aq) —- mno2(s) co2 (aq) [acidic] 2.) calcu…](https://i.ytimg.com/vi/aJ2D0iVT9Eg/mqdefault.jpg) 0:33
0:33
1.) balance this skeleton reaction: mn2 (aq) co3 (aq) —- mno2(s) co2 (aq) [acidic] 2.) calcu…
-
 0:33
0:33
consider the chemical reaction below: zn 2 hcl → zncl2 h2 select the statements that are true…
-
![the ph of a solution is 9.67. calculate the hydrogen ion concentration in the solution. [h ] = 2.1 …](https://i.ytimg.com/vi/HEOFS2_yJak/mqdefault.jpg) 0:33
0:33
the ph of a solution is 9.67. calculate the hydrogen ion concentration in the solution. [h ] = 2.1 …
-
 0:33
0:33
hydrogen gas has several advantages and disadvantages as a potential fuel. hydrogen can be obtained…