track 3: understanding the constraints on clinical trials inclusion criteria, control groups, blind
Published 7 years ago • 65 plays • Length 49:02Download video MP4
Download video MP3
Similar videos
-
 54:01
54:01
track 3: how a therapy gets developed and approved the r&d roadmap and the growing role of patients
-
 1:18
1:18
what are the guidelines for genetic testing?
-
 49:53
49:53
day 2: track 2, understanding clinical trials, speaker: kent thoelke
-
 48:00
48:00
track 3 2 30 clinical trial training pre drug deveopment
-
 3:59
3:59
how can patient organizations be research ready?
-
 48:00
48:00
track 3 2 30 clinical trial training pre drug deveopment
-
 1:00:13
1:00:13
menkes disease with daniel defabio from global genes
-
 12:53
12:53
pathway enrichment analysis - simple explanation!
-
 56:28
56:28
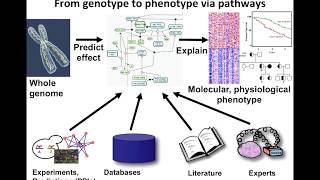
introduction to pathway and network analysis
-
 1:03
1:03
what are the guidelines for genetic testing?
-
 50:32
50:32
track 3: patients as investors
-
 1:03:05
1:03:05
track 3 - patients and caregivers: shaping clinical drug development
-
 33:27
33:27
global genes workshop: introduction to participating in rare clinical trials
-
 51:24
51:24
understanding basket/umbrella trials & master protocols in the context of patient-centered research
-
 1:05:12
1:05:12
track 3 - patients and advocates as entrepreneurs: new models for drug development
-
 1:03:31
1:03:31
rare drug development symposium: advancing diversity & inclusion in clinical research
-
 56:19
56:19
track 3: show your data what do you need to have before approaching industry
-
 1:01:26
1:01:26
track 2: the promise and perils of working with industry lessons from avocacy group experience on a
-
 1:02:45
1:02:45
track 3 - the value of rare disease therapies: patient perspectives needed
-
 58:11
58:11
track 3 1 10 show me the money!
-
 1:58
1:58
next report interview: harsha rajasimha, organization for rare disease in india in usa