two moles of an ideal gas occupy a volume v. the gas expands isothermally and reversibly to a volum…
Published 2 months ago • 1 view plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 0:33
0:33
calc two moles of an ideal gas are compressed in a cylinder at a constant temperature of 65.0^…
-
 0:35
0:35
when 2.0 moles of an ideal gas expands from 5.0l to 10.0 l reversibly and isothermally at 27°....
-
 0:33
0:33
(ii) an ideal gas expands isothermally (t=410 k) from a volume of 2.50 l and a pr…
-
 0:33
0:33
entropy a 2.50 mol sample of an ideal gas expands reversibly and isothermally at 360 k unt…
-
 0:33
0:33
a physics lecture room at 1.00 atm and 27.0â°c has a volume of 216 mâ³. (a) use the ideal gas law t…
-
 6:35
6:35
an ideal gas expands isothermally from a volume \( v_{1} \) to \( v...
-
 0:33
0:33
two moles of an ideal gas have a pressure of 2.0 atm and a temperature of 127 â°c. if the gas is th…
-
 1:17:28
1:17:28
n=2* su(2) supersymmetric yang-mills theory and four-manifold invariants - gregory moore
-
 31:19
31:19
a sum two ways!
-
 10:16
10:16
adu - what size gas line to install? find out
-
 1:22
1:22
one mole of a perfect gas expands isothermally and reversibly to ten times this original volume.....
-
 2:20
2:20
three moles of an ideal gas at 127^(@)c expands isothermally untill the volume is doubled. calcu...
-
 4:06
4:06
(a) one mole of an ideal gas expands isothermally and reversibly at
-
 4:59
4:59
an ideal gas expands isothermally from a volume \( v_{1} \) to \( v...
-
 5:31
5:31
an ideal gas expands isothermally from volume v 1 to v2 and is then compressed to original volume
-
 12:41
12:41
d2 ideal gases
-
 14:06
14:06
two nice double sums!
-
 2:01
2:01
lesson 11 - the ideal gas law, part 2 (chemistry tutor)
-
 23:31
23:31
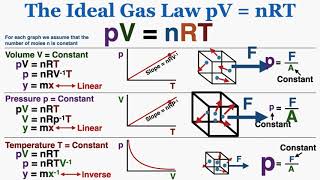
the ideal gas law: pv = nrt - ib physics
-
 9:47
9:47
three moles of an ideal gas being initially at a temperature t_i=273k were isothermally expanded...
-
 4:03
4:03
chemical thermodynamics class 12 chemistry numericals | part 1| estimate amount of work in dm³
-
 0:33
0:33
(i) what potential difference is needed to give a helium nucleus (q=3.2 ×10^-19 …