worked example: calculating solubility from kₛₚ | equilibrium | ap chemistry | khan academy
Published 3 years ago • 50K plays • Length 4:52Download video MP4
Download video MP3
Similar videos
-
 6:27
6:27
calculating ksp value of a salt example 1
-
 4:50
4:50
example: determining which solid will precipitate first (solubility equilibrium #4)
-
 4:24
4:24
calculate the solubility of ag3po4 in (a) pure water. (b) 0.20 mol l-1 na3po4 solution.
-
 4:11
4:11
17.8c using the solubility of a compound to calculate ksp
-
 4:55
4:55
refer to the ksp value from table 17 2 to calculate the solubility of ironii hydroxide in pure water
-
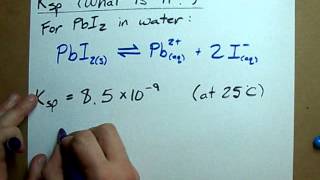 3:40
3:40
what is ksp? (solubility product constant)
-
 4:53
4:53
calculating ksp
-
 6:49
6:49
predicting precipitation with ksp values
-
 4:37
4:37
calculate ksp using one ion concentration
-
![15.28a | calculate the ksp for agbr: [ag ] = 5.7 × 10–7 m, [br–] = 5.7 × 10–7 m](https://i.ytimg.com/vi/eb9YTHd3HcI/mqdefault.jpg) 4:01
4:01
15.28a | calculate the ksp for agbr: [ag ] = 5.7 × 10–7 m, [br–] = 5.7 × 10–7 m
-
 28:39
28:39
chem 1b-lec 34-solubility products (ksp)
-
 6:34
6:34
what mass will dissolve in 1 l of water? (use ksp)
-
 5:26
5:26
determine ksp given solubility
-
 5:16
5:16
example: determining whether a precipitate will form (solubility equilibrium #3)
-
 0:33
0:33
ksp value for lead iodide was determined to be 7.31ã—10^-9. use this information to calculate the s…
-
 2:57
2:57
7.13 solubility problem: ph from ksp
-
 0:33
0:33
1. calculate the ksp value for pbcl2 if 50.0 ml of a saturated solution of pbcl2 has 0.2207 g of pb…
-
 14:58
14:58
experiment 6 solubility calculations
-
 3:14
3:14
chapter 17. calculating ksp from ph or poh