validation of analytical methods according to the latest ich q2(r2) guidelines – part 2
Published 2 months ago • 168 plays • Length 12:01Download video MP4
Download video MP3
Similar videos
-
 10:53
10:53
validation of analytical methods according to new ich q2(r2) guideline
-
 15:30
15:30
general considerations for validation of analytical procedures as per ich guideline q2(r2)
-
 5:24
5:24
ich q2 r1 || analytical method validation || identification test by ir ||
-
 32:41
32:41
performance characteristic: validation of analytical procedures as per ich
-
 30:13
30:13
ich q2 validation of analytical procedures for pharmaceutical total organic carbon analyzers
-
 2:43
2:43
ich q2: guidelines for method validation💥💊 #interview
-
 19:35
19:35
what are the proposed changes in the reportable range as per the draft ich guideline -q2(r2)
-
 27:55
27:55
ich guidelines part-ii;range,accuracy, precision, lod, loq, robustness & system suitability criteria
-
 1:05:33
1:05:33
assay: analytical method validation tutorial: step-by-step with examples #validation #pharma
-
 9:43
9:43
key terms related to validation of an analytical method
-
 13:29
13:29
ich q14-analytical procedure development #pharmatube
-
 8:17
8:17
ich q2r1 analytical method validation
-
 36:25
36:25
ich guidelines for analytical method validation (q2a and q2b); specificity and linearity part- i
-
 2:47
2:47
ich q2: validation of analytical procedures: text and methodology
-
 24:00
24:00
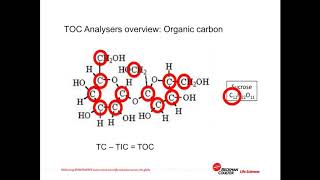
ich q2 validation of on line toc analysers
-
 3:48
3:48
why is analytical method validation required | requirements of analytical method validation
-
 12:26
12:26
what are the differences in method validation between ich and anvisa?
-
 16:58
16:58
why does ich recommend only specificity for the validation of the identification method?
-
 22:58
22:58
are you checking linearity correctly? method validation | ich q2| drawbacks | a new approach
-
 18:23
18:23
validation of analytical method | method validation | validation of an analytical procedure