vcls webinar - opportunities for early dialogue with the ema and hta bodies
Published 6 years ago • 238 plays • Length 1:54Download video MP4
Download video MP3
Similar videos
-
 4:16
4:16
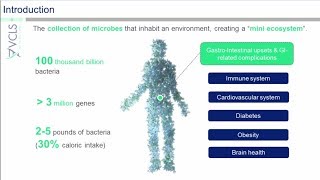
regulation of probiotics: challenges and opportunities
-
 3:22
3:22
vcls webinar - microbiome: navigating regulatory and cmc technical development roadmap
-
 6:51
6:51
the microbiome journey: development of a food product with health claims
-
 2:34
2:34
unlocking the ema meetings: the process and how to prepare adequately
-
 2:22
2:22
achieve success for your us product application
-
 4:17
4:17
microbiome health products: drugs or foods?
-
 2:35
2:35
teaser - how to master marketing authorization & joint clinical assessment 🏥🚀
-
 1:12
1:12
advanced therapy and medicinal products: how to overcome the hurdles from bench to clinic ?
-
 4:01
4:01
to go or not to go into parallel consultation?
-
 1:08
1:08
vcls @ bio europe fall 2017
-
 3:04
3:04
teaser how to secure early stage development and first in human studies
-
 0:30
0:30
vcls in one word
-
 1:15
1:15
pharma pricing & market access congress