wavelength of light released from hydrogen
Published 11 years ago • 131K plays • Length 4:56Download video MP4
Download video MP3
Similar videos
-
 21:44
21:44
bohr model of the hydrogen atom, electron transitions, atomic energy levels, lyman & balmer series
-
 2:54
2:54
calculate the wavelength of the photon emitted when the hydrogen atom transition from n=5 to n=3.
-
 2:30
2:30
atomic structure - finding the wavelength of light to excite a hydrogen electron
-
 5:36
5:36
what is the wavelength of light emitted when the electron in a hydrogen atom undergoes transition
-
 3:51
3:51
energy released: δe for electron in hydrogen atom
-
 8:19
8:19
calculate emission wavelength and frequency from change in energy levels - visible series 001
-
 7:36
7:36
wavelength of light from energy (1 of 2)
-
 4:10
4:10
⚗️ wavelength of light for a transition in the hydrogen atom (question 2)
-
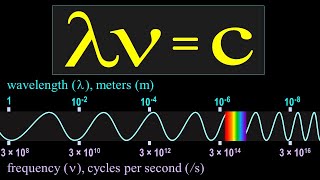 9:27
9:27
frequency, wavelength, and the speed of light
-
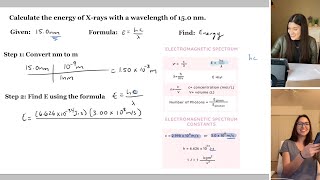 36:02
36:02
how to find the wavelength, frequency, energy and photons | study chemistry with us
-
 16:49
16:49
grade 10 science | quarter 2 module 2 part 1 | em wave calculations (wavelength and frequency)
-
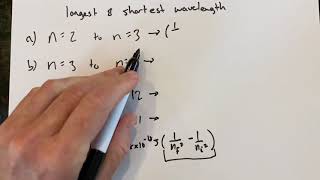 6:40
6:40
hydrogen transitions and wavelength
-
 5:18
5:18
wavelength of light from energy (2 of 2)
-
 6:01
6:01
⚗️ wavelength of light for a transition in the hydrogen atom (question 3)
-
 9:37
9:37
calculate emission wavelength and frequency from change in energy levels 002
-
 3:24
3:24
light of wavelength 12818å is emitted when the electron of a hydrogen atom drops
-
 5:41
5:41
a monochromatic radiation of wavelength 975 å excites the hydrogen atom from its ground state to a h
-
 3:35
3:35
calculate the wavelength, in nanometers,of the spectral line produced when an electron in a hydrogen
-
 2:05
2:05
calculate the frequency and the wavelength of the light emitted when the electron in a hydrogen a...
-
 5:55
5:55
⚗️ wavelength of light for a transition in the hydrogen atom (question 1)
-
 3:50
3:50
calculating wavelengths of emitted light in the hydrogen spectrum
-
 2:41
2:41
a photon of energy 10.2 ev corresponds to light of wavelength `lamda_(0)`. due to an