what do fda reviewers look for in 510 (k) and ide submissions?
Published 9 years ago • 1.2K plays • Length 2:41Download video MP4
Download video MP3
Similar videos
-
 2:16
2:16
concerns about 510(k) process
-
 2:32
2:32
what were the most common submission mistakes you saw as an fda lead reviewer?
-
 2:35
2:35
perclose: 510(k) vs. pma
-
 0:52
0:52
510(k) and market potential
-
 2:51
2:51
key differences between 510(k) and pma pathways at the fda
-
 9:30
9:30
fda estar template has changed 10 things
-
 1:34:30
1:34:30
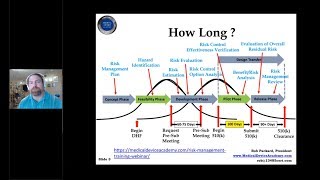
how to prepare a medical device 510k submission for fda | rob packard | joe hage
-
 27:53
27:53
top of the line: fiio fd7 first look!
-
 2:39
2:39
cms and fda
-
 1:37
1:37
structure of fda/cdrh
-
 2:56
2:56
ce or us fda 510(k): what should you choose for your medical device?
-
 4:11
4:11
what is the difference between a 510k and de novo?
-
 0:17
0:17
510(k) tip - make sure your ifu template includes all of the fda estar requirements.
-
 2:19
2:19
understanding the fda medical device 510k process
-
 0:38
0:38
510(k) tip - try filling in the fda estar template while you are obtaining testing quotes
-
 2:06
2:06
fda reviews medical device approval process
-
 4:03
4:03
did you know? find 510k by indications for use
-
 4:25
4:25
understanding the difference between fda approval and fda clearance
-
 0:14
0:14
determining branch at fda
-
 3:31
3:31
510k submission | integrated assessment services (ias)
-
 0:56
0:56
check out our new fda us agent website
-
 22:34
22:34
fda establishment registration and listing for medical devices