what is a class 1 and 2 device exemption?
Published Streamed 1 year ago • 1K plays • Length 12:06Download video MP4
Download video MP3
Similar videos
-
 1:27
1:27
investigational device exemption
-
 2:18
2:18
what is an investigational device exemption (ide)?
-
 4:11
4:11
what is the difference between a 510k and de novo?
-
 10:59
10:59
medical devices classification as per fda | medical device regulations | #medicaldevices #fda
-
 3:49
3:49
are you mdsap? - the medical device single audit program for medical device industry
-
 10:39
10:39
product development 101 // medical device startup guide
-
 9:28
9:28
medical device regulations / fda approval
-
 18:53
18:53
what is the regulatory pathway for a de novo medical device or ivd?
-
 26:21
26:21
medical devices class i under mdr 2017/745 with erik vollebregt (part 2)
-
 0:33
0:33
what does it mean to be a class ii medical device?
-
 16:01
16:01
do you have a procedure for release of product to a clinical study?
-
 2:30
2:30
role of investigational device exemption (ide)
-
 6:34
6:34
510(k) submission predicate selection webinar - updated for 2022
-
 16:38
16:38
do you know the difference between a medical device component and an accessory medical device?
-
 0:16
0:16
doctor vs. nurse: $ over 5 years #shorts
-
 25:01
25:01
what are the four different types of medical device risk analysis?
-
![regulatory pathway: reach the us market [class ii 510k exempt]](https://i.ytimg.com/vi/BWK7LQPt8ZY/mqdefault.jpg) 32:50
32:50
regulatory pathway: reach the us market [class ii 510k exempt]
-
 1:26
1:26
medical device product classification
-
 0:14
0:14
highest paid healthcare workers 💰 (that aren't medical doctors) #shorts
-
 11:24
11:24
medical device classification overview
-
 19:08
19:08
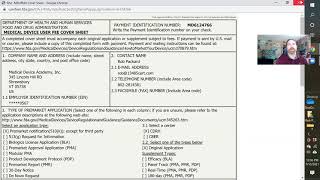
510(k) estar webinar - indications for use and classification
-
 22:34
22:34
fda establishment registration and listing for medical devices