what is investigational new drug (ind) application? | regulatory learnings | drug regulatory affairs
Published 2 years ago • 8.4K plays • Length 5:30Download video MP4
Download video MP3
Similar videos
-
 4:25
4:25
step-wise approach for investigational new drug (ind) application? | regulatory learnings | dra
-
 2:15
2:15
step 6: how does usfda review an investigational new drug (ind) application? | regulatory learnings
-
 8:54
8:54
investigational new drug(ind)
-
 46:00
46:00
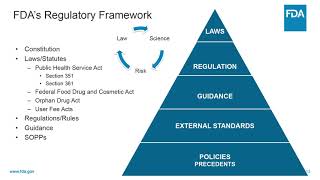
introduction to investigational new drug (ind) applications (3/14) redi 2017
-
 28:23
28:23
fda product regulations part 1 of 7
-
 1:00:47
1:00:47
understanding new drug applications (ndas)
-
 36:32
36:32
lecture 2- investigational new drug (ind) application (part-1) (unit-3) by payal n. vaja
-
 3:59
3:59
step 5: how to submit an investigational new drug (ind) application to usfda? | regulatory learnings
-
 6:46
6:46
investigational new drug application: key to starting clinical trials | regulatory affairs
-
 10:36
10:36
investigational new drug | ind | pharmaceutical regulatory affairs | m.pharm
-
 6:30
6:30
regulatory shorts#7 | patent vs data exclusivity vs market exclusivity | drug regulatory affairs
-
 3:25
3:25
how to register an investigational new drug (ind) to the us fda
-
 3:13
3:13
step 6: how does usfda review an investigational new drug (ind) application (part 2)? | dra
-
 7:21
7:21
step 1: how to prepare the regulatory strategies for ind application? | regulatory learnings | dra
-
 1:11
1:11
investigational new drug (ind) application
-
 16:32
16:32
investigational new drug (ind)
-
 8:17
8:17
step 4: what content do we include in ind application (part 1)? | regulatory learnings | dra
-
 4:17
4:17
regulatory shorts#2 | dsur vs psur | safety reports | drug regulatory affairs
-
 2:04
2:04
investigational new drug application inda - lesson on learners' request
-
 1:15:32
1:15:32
webinar about us investigational new drug (ind) applications
-
 5:52
5:52
step 2: what is target product profile (tpp)? | regulatory learnings | drug regulatory affairs