6.36 | which of the subshells described in the previous question contain degenerate orbitals? how
Published 4 years ago • 4.6K plays • Length 6:34
Download video MP4
Download video MP3
Similar videos
-
 9:23
9:23
shells, subshells, and orbitals - biology/chemistry ep5
-
 16:11
16:11
6.41 | consider the orbitals shown here in outline. (a) what is the maximum number of electrons
-
 11:19
11:19
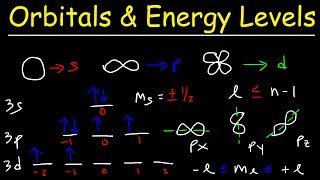
orbitals, atomic energy levels, & sublevels explained - basic introduction to quantum numbers
-
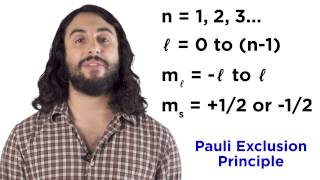 8:42
8:42
quantum numbers, atomic orbitals, and electron configurations
-
 0:48
0:48
how small are atoms?
-
 8:43
8:43
6.66 | write a set of quantum numbers for each of the electrons with an n of 3 in a sc atom.
-
 5:35
5:35
a better way to picture atoms
-
 21:34
21:34
what are atomic orbitals?
-
 5:50
5:50
atomic orbitals 3d
-
 6:37
6:37
6.79 | which atom and/or ion is (are) isoelectronic with br : se2 , se, as-, kr, ga3 , cl-?
-
 4:06
4:06
6.37 | identify the subshell in which electrons with the following quantum numbers are found
-
 6:00
6:00
what are shells, subshells, and orbitals? | chemistry
-
 14:30
14:30
6.53 | use an orbital diagram to describe the electron configuration of the valence shell of each of
-
 10:10
10:10
8.41a | how to find the bond orders of h2, h2 , and h2 -
-
 19:19
19:19
6.46 | read the labels of several commercial products and identify monatomic ions of at least four
-
 11:26
11:26
6.52 | draw the orbital diagram for the valence shell of each of the following atoms: (a) c (b) p
-
 10:52
10:52
orbitals: crash course chemistry #25
-
 0:22
0:22
atom 3d model orbital spin
-
 5:20
5:20
what are degenerate orbitals according to hunds rule, how are degenerate orbitals occupied
-
 9:23
9:23
6.26 | using the bohr model, determine the energy in joules of the photon produced when an electron
-
 5:13
5:13
6.57 | which ion with a 1 charge has the electron configuration 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6?
Clip.africa.com - Privacy-policy
 9:23
9:23
 16:11
16:11
 11:19
11:19
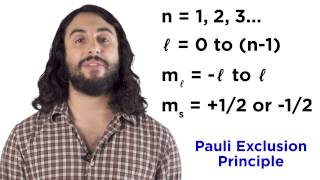 8:42
8:42
 0:48
0:48
 8:43
8:43
 5:35
5:35
 21:34
21:34
 5:50
5:50
 6:37
6:37
 4:06
4:06
 6:00
6:00
 14:30
14:30
 10:10
10:10
 19:19
19:19
 11:26
11:26
 10:52
10:52
 0:22
0:22
 5:20
5:20
 9:23
9:23
 5:13
5:13