biosimilar medications — what patients need to know
Published 1 year ago • 4.8K plays • Length 3:02Download video MP4
Download video MP3
Similar videos
-
 1:37
1:37
biosimilar medications – what health care providers need to know
-
 1:58
1:58
fda review and approval process for biosimilar medications
-
 4:12
4:12
the basics of biosimilars
-
 0:31
0:31
biosimilar medications: as identical twins explain, biosimilars have the same benefits (30 sec.)
-
 2:47
2:47
biosimilars: approval process
-
 3:07
3:07
the promise of biosimilars
-
 5:20
5:20
data requirements for biosimilars
-
 4:58
4:58
overview of the toxicities associated with bispecific agents in multiple myeloma
-
 1:36
1:36
interchangeable biosimilars
-
 59:48
59:48
generic drugs and biosimilars 101
-
 22:48
22:48
navigating the world of biosimilar user fees- june 10, 2019
-
 0:16
0:16
biosimilar medications: as identical twins explain, biosimilars have the same benefits (15 sec.)
-
 14:31
14:31
biosimilars and interchangeables - regulatory highlights (27of33) quality – oct. 16-17, 2019
-
 7:21
7:21
biologic drugs and biosimilars
-
 39:51
39:51
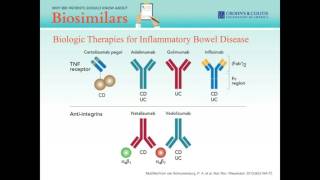
why ibd patients should know about biosimilars
-
 25:07
25:07
office of clinical pharmacology (ocp): biosimilars - bioanalysis 2020
-
 51:06
51:06
biosimilars and oncology clinical pathways: perfect together
-
 3:03
3:03
what physicians need to know to safely prescribe biosimilars
-
 11:18
11:18
biosimilar and interchangeable insulins: what clinicians need to know