fda review and approval process for biosimilar medications
Published 1 year ago • 3.7K plays • Length 1:58Download video MP4
Download video MP3
Similar videos
-
 2:47
2:47
biosimilars: approval process
-
 3:03
3:03
biosimilar medications — what patients need to know
-
 4:12
4:12
the basics of biosimilars
-
 8:31
8:31
fda d.i.s.c.o.: first biosimilar approval for the treatment of cancer
-
 22:48
22:48
navigating the world of biosimilar user fees- june 10, 2019
-
 4:20
4:20
the biosimilar development process
-
 2:02
2:02
5 things you need to know about the drug approval process
-
 1:31:06
1:31:06
the fda drug development process: glp, gmp and gcp regulations
-
 32:37
32:37
biologics, biosimilars, and the two-fold u.s. approval framework’s possible impact on prices
-
 11:12
11:12
how i brought my first product to market – idea to launch
-
 1:36
1:36
interchangeable biosimilars
-
 5:20
5:20
data requirements for biosimilars
-
 0:31
0:31
biosimilar medications: as identical twins explain, biosimilars have the same benefits (30 sec.)
-
 24:48
24:48
fda perspectives on biosimilar bla-manufacturing (28of33) quality – oct. 16-17, 2019
-
 22:45
22:45
process for reviewing nonproprietary name suffix for biological products – pharmacovigilance 2020
-
 8:17
8:17
fda review and approval of drugs and medical devices
-
 42:43
42:43
regulatory highlights for biosimilars and interchangeables (9of15) redi – may 29-30, 2019
-
 14:31
14:31
biosimilars and interchangeables - regulatory highlights (27of33) quality – oct. 16-17, 2019
-
 36:55
36:55
components of new drug application and biologics license application (5of15) redi– may 29-30, 2019
-
 54:40
54:40
otc monograph reform overview of draft guidance for formal meetings
-
 14:19
14:19
regulatory pathway for biosimilars
-
 3:33
3:33
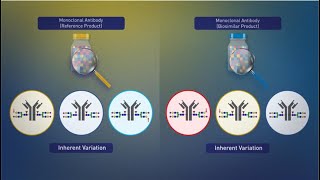
biosimilars: manufacturing and inherent variation