clinical trials in emergency situations
Published 9 months ago • 116 plays • Length 20:15Download video MP4
Download video MP3
Similar videos
-
 2:01
2:01
clinical trials in the eu - en version
-
 0:31
0:31
clinical trials in the eu - short version
-
 5:06
5:06
ctis - m02 ctis common functionalities part a
-
 16:00
16:00
implementation of the clinical trials regulation
-
 42:31
42:31
overview of the european medicines agency (ema), part 1 of 3
-
 2:28
2:28
what is the chmp?
-
 2:05
2:05
accelerating clinical trials in the eu (act eu)
-
 1:03:18
1:03:18
understanding europe's medical device regulation
-
 6:52
6:52
independent interpretation
-
 57:40
57:40
eu clinical trials regulation – challenges drug developers faced in the first 6 months
-
 1:00
1:00
act now: transition your clinical trials to ctis | important information for clinical trial sponsors
-
 18:54
18:54
accelerating clinical trials in the eu (act-eu) by i. den rooijen (ema)
-
 1:58
1:58
what ema does for public and animal health
-
 1:54:36
1:54:36
webinar on revision of the pharmaceutical legislation
-
 2:39
2:39
how does ema ensure the safety of medicines in the eu
-
 6:39
6:39
ctis – m08 how to assess an initial clinical trial application in ctis – part i
-
 4:52
4:52
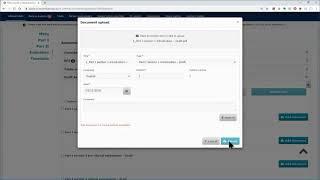
ctis – m10 how to submit an initial cta in the ctis – fill in the form and the msc sections.
-
 1:07:39
1:07:39
webinar on transparency rules for the eu clinical trials information system ctis
-
 12:27
12:27
#episode10 - european medicines agency (ema) : the darwin eu project
-
 2:13
2:13
clinical trial regulation: european medicines agency
-
 1:46
1:46
european medicines agency on astrazeneca: ‘this is a safe and effective vaccine’ | nbc news now
-
 23:44
23:44
ema press briefing 21 october 2021