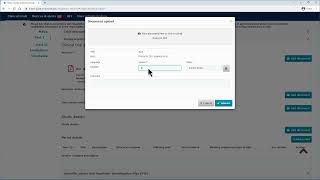
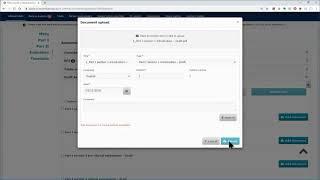
ctis – m10 how to submit an initial cta in the ctis – fill in the form and the msc sections.
Published 3 years ago • 13K plays • Length 4:52Download video MP4
Download video MP3
Similar videos
-
 10:03
10:03
ctis – m10 how to submit an initial cta in the ctis sponsor workspace – fill in the part i section
-
 3:35
3:35
ctis – m10 how to submit an initial cta in the ctis – fill in the product details of part i section.
-
 4:14
4:14
ctis – m10 how to submit an initial cta in the ctis – fill in the trial details of part i section.
-
 5:20
5:20
ctis – m10 how to submit an initial cta in the ctis – fill in the part ii section
-
 4:00
4:00
ctis – m10 how to submit an initial cta in the ctis – fill in the sponsor details of part i section.
-
 3:41
3:41
ctis – m10 how to submit an additional msc application in the ctis sponsor workspace
-
 7:43
7:43
ctis – m10 how to submit a single trial substantial modification in the ctis sponsor workspace
-
 6:39
6:39
ctis – m08 how to assess an initial clinical trial application in ctis – part i
-
 5:38
5:38
ctis – m11 how to change a clinical trial application as part of an rfi response (sponsors)
-
 1:29:43
1:29:43
clinical trials information system ctis bitesize talk: initial clinical trial application
-
 1:28:16
1:28:16
1st dadi eaf training webinar
-
 4:43
4:43
ctis – m08 how to assess an initial clinical trial application in ctis – part ii
-
 3:25
3:25
ctis - m06 how to access an initial cta in ctis – validation - rfi and issue validation decision
-
 7:34
7:34
ctis - m20 circulate draft asr - ar, create and consolidate considerations
-
 5:38
5:38
ctis - m06 how to access an initial cta in ctis – validation - document considerations
-
 3:22
3:22
ctis - m03 registration of a new organisation in ctis
-
 57:25
57:25
ctis walk in clinic
-
 44:19
44:19
ctis walk in clinic
-
 5:06
5:06
ctis - m02 ctis common functionalities part a
-
 4:56
4:56
ctis - m17 how to create an ad hoc assessment – discussion with mss and create rfis
-
 5:30
5:30
ctis - m06 how to access an initial cta in ctis – rms selection