edited version - webinar about us investigational new drug (ind) applications.
Published 3 years ago • 344 plays • Length 1:12:07Download video MP4
Download video MP3
Similar videos
-
 1:15:32
1:15:32
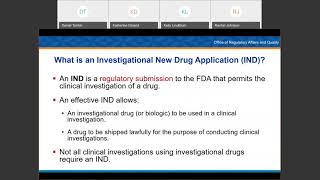
webinar about us investigational new drug (ind) applications
-
 46:00
46:00
introduction to investigational new drug (ind) applications (3/14) redi 2017
-
 40:34
40:34
demystifying the investigational new drug (ind) application for drugs and biologics (3of14) redi '18
-
 5:30
5:30
what is investigational new drug (ind) application? | regulatory learnings | drug regulatory affairs
-
 3:25
3:25
how to register an investigational new drug (ind) to the us fda
-
 33:18
33:18
investigational new drug (ind) submission: content/format and first 30 days (5of14) redi 2018
-
 36:32
36:32
lecture 2- investigational new drug (ind) application (part-1) (unit-3) by payal n. vaja
-
 40:24
40:24
fda communication during drug development (4/14) redi 2017
-
 30:32
30:32
abbreviated new drug application
-
 1:20
1:20
investigational new drug (ind) for us fda | phase 1 to 4 clinical trials services | drug development
-
 2:20:19
2:20:19
fda regulatory affairs webinar - asphalion
-
 3:13
3:13
step 6: how does usfda review an investigational new drug (ind) application (part 2)? | dra
-
 40:53
40:53
submit your investigational new drug (ind) application and clinical holds (9/14) redi 2017
-
 2:15
2:15
step 6: how does usfda review an investigational new drug (ind) application? | regulatory learnings
-
 1:11
1:11
investigational new drug (ind) application
-
 2:03:01
2:03:01
investigational new drug workshop
-
 0:56
0:56
🥰understanding the investigational new drug ind application process#shorts
-
 58:49
58:49
investigational new drug application (ind) forms: updates and best practices
-
 36:36
36:36
heal biomarker webinar
-
 21:20
21:20
investigator responsibilities - safety reporting for investigational drugs and devices