for the reaction, n2 3h2⇌2nh3,δh= ?
Published 18 hours ago • No plays • Length 1:22Download video MP4
Download video MP3
Similar videos
-
 1:30
1:30
for the reaction, n2 3h2→2nh3, if dnh3dt=2×10-4 mol l-1s-1, the value of -dh2dt would be
-
 5:03
5:03
for the reversible reaction, n2( g) 3h2( g)⇌2nh3( g) heat. the equilibrium ....
-
![consider the following reaction n2 3h2— 2nh3 the rate of reaction in terms of n2 is -d[n2]/dt = 0.02](https://i.ytimg.com/vi/smQ8fuXlxtw/mqdefault.jpg) 3:26
3:26
consider the following reaction n2 3h2— 2nh3 the rate of reaction in terms of n2 is -d[n2]/dt = 0.02
-
 2:10
2:10
for the reaction n2 3h2 gives 2nh3, kc depends on
-
 1:16
1:16
consider the reaction 2nh3(g) → n2(g) 3h2(g)
-
 20:12
20:12
stoichiometry - limiting & excess reactant, theoretical & percent yield - chemistry
-
 13:56
13:56
form 5 (chapter 2.3.1(ii))
-
 10:54
10:54
two reactions (extent of reaction method)
-
 1:39
1:39
the equilibrium constant for the reaction, n2 3h2 ⇌ 2nh3 is kc
-
 5:44
5:44
kckp for the reaction, n2(g) 3h2(g) ⇌ 2nh3(g) /11th /physical and chemical equilibrium/ in tamil
-
 2:01
2:01
oqv no – 255 what is the unit of kp for the reversible reaction n2(g) 3h2(g) ↔ 2nh3(g)?
-
 0:36
0:36
for the reaction, n2 3h2 — 2nh3, del h = ?
-
 7:16
7:16
for the following reaction n2 3h2-----* 2nh3! neet 2019 solved chemistry questions in english,allwyn
-
 1:18
1:18
for the chemical reaction, n2(g) 3h2(g)⇌2nh3(g) the correct option is
-
 0:36
0:36
for the chemical reaction, n2 3h2 = 2nh3 the correct option is
-
 0:49
0:49
homogeneous reaction n2(g) 3h2(g) → 2nh3(g). #chemistry
-
 0:56
0:56
determination of the unit of kp for the reaction n2(g) 3h2(g) ↔ 2nh3(g). #chemistry
-
 0:33
0:33
for the reaction 3h2(g) n2(g) ⇜ 2nh3(g) at 225 â°c, the equilibrium constant is 1.7 ã— 10^2. if…
-
 1:23
1:23
the reaction, n2 3h2 → 2nh3 is used to produce ammonia.
-
 1:40
1:40
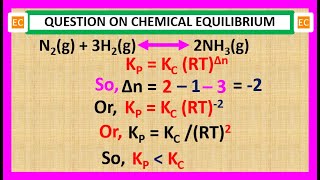
oqv no – 36 relation between kp and kc for the reaction n2 3h2 = 2nh3.
-
 2:35
2:35
13.24a | does kc equal kp for the reaction: n2(g) 3h2(g) ⇌ 2nh3(g)
-
 1:41
1:41
for a chemical reaction,n2(g) 3h2(g)#8652;2nh3(g), the correct option is:....