how many electrons in an atom may have the quantum numbers?(a) n = 4, ms = – ½ (b) n = 3, l = 0
Published 3 years ago • 761 plays • Length 5:03
Download video MP4
Download video MP3
Similar videos
-
 4:30
4:30
how many electrons in an atom may have the following quantum numbers? a) n=4 m=-1/2 b) n=3 l=0
-
 4:04
4:04
how many electrons in an atom may have the following quantum numbers ?
-
 4:15
4:15
how many electrons in an atom may have the following quantum numbers i)n-4, ms=-1/2 ii)n=3,l=0
-
 7:33
7:33
how many electrons in an atom may have the following quantum numbers ? (a) n = 4, ms = -1/2
-
 11:46
11:46
how to determine the maximum number of electrons using allowed quantum numbers - 8 cases
-
 12:12
12:12
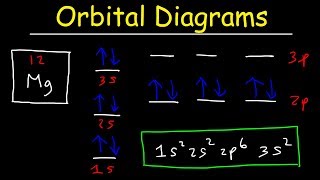
orbital diagrams and electron configuration - basic introduction - chemistry practice problems
-
 10:19
10:19
electron configuration - basic introduction
-
 9:23
9:23
shells, subshells, and orbitals - biology/chemistry ep5
-
 3:04
3:04
how many electron in an atom may have the following quantum number ? a. `n = 4, m_(s) = -(1)/(2)...
-
 8:42
8:42
quantum numbers, atomic orbitals, and electron configurations
-
 2:03:21
2:03:21
class 11 chemistry - structure of atom part 2, quantum numbers, aufbau, hunds, paulis principal.
-
 1:01
1:01
how many electrons in an atom may have the following quantum numbers.
-
 3:47
3:47
, how many electrons in an atom may have the following quantum numbers? (a) n=4, ms=-1/2 (b) n=3...
-
 7:20
7:20
quantum numbers - how many electrons and orbitals have the following set of quantum numbers?
-
 3:46
3:46
how many orbitals are there in n=3, the 3rd energy level of an atom?
-
 4:25
4:25
how to determine the 4 quantum numbers from an element or a valence electron
-
 12:57
12:57
how many electrons in an atom could have these sets of quantum numbers
-
 2:51
2:51
in an atom, the total number of electrons having quantum numbers n=4,| m_l|=1 and m_s=-1 / 2 is
-
 3:00
3:00
how many electrons in an atom may have the following quantum numbers, ncert solutions atomic
-
 47:51
47:51
quantum numbers - n, l, ml, ms & spdf orbitals
-
 1:55
1:55
neet chemistry mcq atomic structure 92
-
 1:01
1:01
which of the following sets of quantum numbers is correct for an electron in 4f orbital jee mains 04
Clip.africa.com - Privacy-policy
 4:30
4:30
 4:04
4:04
 4:15
4:15
 7:33
7:33
 11:46
11:46
 12:12
12:12
 10:19
10:19
 9:23
9:23
 3:04
3:04
 8:42
8:42
 2:03:21
2:03:21
 1:01
1:01
 3:47
3:47
 7:20
7:20
 3:46
3:46
 4:25
4:25
 12:57
12:57
 2:51
2:51
 3:00
3:00
 47:51
47:51
 1:55
1:55
 1:01
1:01