how to prepare an fda estar 510(k) submission
Published 3 years ago • 4.3K plays • Length 38:26Download video MP4
Download video MP3
Similar videos
-
 37:47
37:47
using the new estar templates for a 510(k) submission and the fda estar draft guidance
-
 17:20
17:20
fda estar - general info - #1 in the 510(k) estar series
-
 11:06
11:06
how to prepare a medical device 510k submission for fda
-
 6:34
6:34
510(k) submission predicate selection webinar - updated for 2022
-
 11:30
11:30
finally fda cdrh releases an fda estar draft guidance for the estar 510(k) templates
-
 12:15
12:15
510(k) tips and tricks
-
 1:34:30
1:34:30
how to prepare a medical device 510k submission for fda | rob packard | joe hage
-
 1:15:28
1:15:28
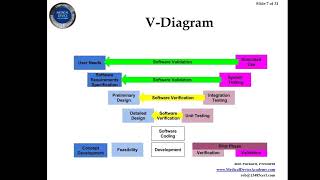
how to you create a design history file (dhf)?
-
 1:19:59
1:19:59
requirements contents and options : the 510k submission
-
 1:51:45
1:51:45
as13100 key requirements for process fmea & control plans webinar series – session 1
-
 0:16
0:16
510(k) tip - saving your work
-
 0:57
0:57
using clinical data in your 510(k) fda estar - webinar feb. 8, 2024
-
 52:43
52:43
webinar for special 510(k) submissions
-
 9:30
9:30
fda estar template has changed 10 things
-
 1:12:32
1:12:32
rta hold and ai request responses to an fda 510(k) reviewer
-
 1:06:28
1:06:28
mastering your 510(k) submission process
-
 12:48
12:48
preparing an fda 510 (k) submission often called 510k
-
 19:08
19:08
510(k) estar webinar - indications for use and classification
-
 13:40
13:40
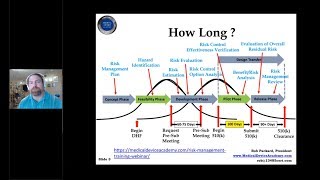
how long does a 510(k) submission take to get cleared?
-
 2:45
2:45
changes to the fda ecopy submission process