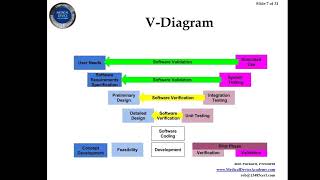
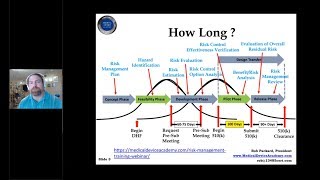
using the new estar templates for a 510(k) submission and the fda estar draft guidance
Published 3 years ago • 4.6K plays • Length 37:47Download video MP4
Download video MP3
Similar videos
-
 11:30
11:30
finally fda cdrh releases an fda estar draft guidance for the estar 510(k) templates
-
 38:26
38:26
how to prepare an fda estar 510(k) submission
-
 17:20
17:20
fda estar - general info - #1 in the 510(k) estar series
-
 6:34
6:34
510(k) submission predicate selection webinar - updated for 2022
-
 0:57
0:57
using clinical data in your 510(k) fda estar - webinar feb. 8, 2024
-
 55:14
55:14
walkthrough of atera’s solution for it departments
-
 15:35
15:35
what is a de novo?
-
 1:15:28
1:15:28
how to you create a design history file (dhf)?
-
 19:08
19:08
510(k) estar webinar - indications for use and classification
-
 11:06
11:06
how to prepare a medical device 510k submission for fda
-
 9:30
9:30
fda estar template has changed 10 things
-
 7:48
7:48
health canada pilot with the fda estar template
-
 7:28
7:28
now you can upload an fda estar or ecopy using the fda customer collaboration portal (ccp)
-
 0:16
0:16
510(k) tip - update your user manual template to include all labeling elements in the fda estar
-
 25:33
25:33
fda estar draft guidance document released september 29, 2021
-
 0:16
0:16
510(k) tip - saving your work
-
 15:04
15:04
cybersecurity documentation for a 510(k) submission
-
 48:19
48:19
fda's 510(k) modernization: what if anything has changed? is changing? will change?
-
 0:53
0:53
510(k) tip - make sure you are signed up for email notifications of new fda guidance documents
-
 16:50
16:50
important new fda guidance – coming soon
-
 1:34:30
1:34:30
how to prepare a medical device 510k submission for fda | rob packard | joe hage