investigational device exemption workshop
Published 3 years ago • 1.7K plays • Length 1:58:39Download video MP4
Download video MP3
Similar videos
-
 1:27
1:27
investigational device exemption
-
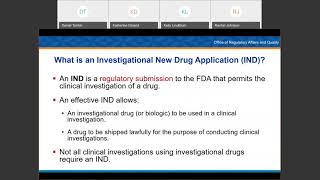 2:03:01
2:03:01
investigational new drug workshop
-
 2:30
2:30
role of investigational device exemption (ide)
-
 2:18
2:18
what is an investigational device exemption (ide)?
-
 2:13:52
2:13:52
oraq ide workshop - june 14, 2017
-
 1:55:08
1:55:08
oraq ind workshop
-
 58:49
58:49
investigational new drug application (ind) forms: updates and best practices
-
 3:08
3:08
duke students use 3d metal printer to create medical devices
-
 15:35
15:35
what is a de novo?
-
 16:38
16:38
do you know the difference between a medical device component and an accessory medical device?
-
 56:55
56:55
fda process for medical device startups: an investor's point of view
-
 54:15
54:15
fda oversight of tobacco product investigations – itp or ind?
-
 1:11:12
1:11:12
the medical device ecosystem through dcri-fda public private partnerships
-
 3:43
3:43
medtronic ceo omar ishrak on data in healthcare
-
 2:45:59
2:45:59
oraq ind workshop - june 13, 2017
-
 2:20
2:20
unc and duke creating new triangle research center to work with fda
-
 12:06
12:06
what is a class 1 and 2 device exemption?
-
 1:12
1:12
these students are 3d-printing biomedical devices
-
 1:32
1:32
duke hospitals to use weapons detection system
-
 10:15
10:15
episode 4: review and update of device establishment inspection processes and standards
-
 0:54
0:54
part 2 | what is the impact of cardiac implantable electronic device infection?