looking for the best and the brightest to join fda’s center for drug evaluation and research
Published 2 years ago • 3.4K plays • Length 2:42Download video MP4
Download video MP3
Similar videos
-
 3:40
3:40
many just need an opportunity: join fda’s center for drug evaluation and research (cder)
-
 0:30
0:30
how does the fda approve new drugs? (30 seconds)
-
 13:21
13:21
overview of the fda product-specific guidance program
-
 0:30
0:30
what does the fda do after drugs are approved? (30 seconds)
-
 5:20
5:20
data requirements for biosimilars
-
 1:08:18
1:08:18
how fda trains its investigators to review capa and what you should do to prepare
-
 4:11
4:11
why does the fda exist?
-
 24:20
24:20
overview of the quality system regulation
-
 5:00
5:00
who is the u.s. food and drug administration fda
-
 0:41
0:41
the fda allows this?! 🤮
-
 1:49
1:49
fda fails to inspect thousands of drug factories
-
 7:30
7:30
what is the software precertification (pre-cert) pilot program?
-
 39:09
39:09
walkthrough of an fda clinical investigator site inspection (12/14) redi 2017
-
 0:30
0:30
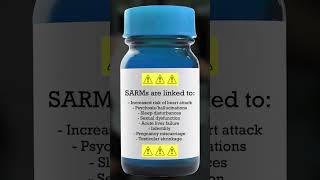
are you thinking about taking sarms? fda warns of sarms use among teens, young adults
-
 3:44
3:44
fda cder regulatory science: the importance of partnership and consortia
-
 10:57
10:57
list of relevant quality guidances & common deficiencies observed during dmf review
-
 14:52
14:52
postmarketing safety and surveillance of generic drugs update
-
 4:11
4:11
the american chamber of horrors
-
 8:58:15
8:58:15
fda regulatory education for industry (redi) – devices and biologics track
-
 1:58
1:58
fda review and approval process for biosimilar medications
-
 24:56
24:56
api facility inspections