medical devices: how to safeguard patients and innovate
Published 10 years ago • 329 plays • Length 2:11Download video MP4
Download video MP3
Similar videos
-
 1:28
1:28
medical devices - an introduction to the eu eudamed system
-
 2:43
2:43
the uk vs the eu medical device regulations - what are the key differences?
-
 3:34
3:34
medical devices: better regulation for europe
-
 1:00:00
1:00:00
webinar | safer medical devices: implementation of the eu mdr and eudamed database
-
 12:41
12:41
medical device security: a thing smart people do | avi rubin | tedxemory
-
 12:14
12:14
first-to-market medical device sparks tense negotiations | shark tank aus
-
 16:04
16:04
introduction european medical device regulation
-
 1:25:47
1:25:47
eu postmarket surveillance requirements for medical devices
-
 37:29
37:29
#cybersecurity for medical devices with erik vollebregt (mdr & ivdr)
-
 5:46
5:46
what is a medical device?
-
 1:02:07
1:02:07
live: ubc news tonight @10pm | august 31, 2024
-
 7:13
7:13
regulatory pathways of medical devices in usa and european union
-
 52:19
52:19
changes in europe’s medical device regulations: consequences for manufacturers
-
 3:56
3:56
medical device innovation - the non-injectable arterial connector (nic)
-
 3:01
3:01
one of a kind medical devices... and how eu funding makes them a reality
-
 16:18
16:18
how is my medical device classified?
-
 13:56
13:56
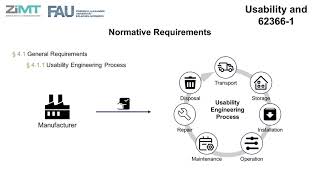
usability engineering in the medical device industry in the european union
-
 14:55
14:55
short course on the medical device regulation (eu) 2017/745
-
 5:33
5:33
medical device innovation - venner pneux tm
-
 12:22
12:22
benefit-risk requirements in eu-mdr
-
 48:15
48:15
how to choose your prrc ? (eu mdr 2017/745 - ivdr 2017/746)
-
 20:38
20:38
how to deal with significant changes for your medical devices (eu mdr)