overview of the current medical device regulatory framework in singapore part 2 of 2
Published 11 years ago • 2.3K plays • Length 9:35Download video MP4
Download video MP3
Similar videos
-
 8:39
8:39
overview of the current medical device regulatory framework in singapore part 1 of 2
-
 17:49
17:49
fda regulation of medical device software (part 2 of 3)
-
 3:25
3:25
united states medical device registration chapter 3 - quality management system
-
 2:58
2:58
overview of medical device registration
-
 14:55
14:55
short course on the medical device regulation (eu) 2017/745
-
 25:56
25:56
regulatory affairs: how to handle the medical device regulation (mdr)
-
 24:20
24:20
overview of the quality system regulation
-
 22:07
22:07
how to work in regulatory affairs (drug and medical devices)
-
 54:15
54:15
medical device regulation (fda)
-
 28:22
28:22
innovator implications - fda regulation of medical device software (part 3 of 3)
-
 44:52
44:52
medical devices regulation overview in the mena region with baraa nofal
-
 9:01
9:01
demystifying medical device regulatory frameworks
-
 3:12
3:12
prepare to register a medical device: group the products
-
 41:01
41:01
3 systems of medical device risk from fda
-
 32:56
32:56
medical devices regulations webinar - 24 january 2023
-
 45:23
45:23
regulatory gap analysis of fda's framework for medical devices
-
 2:11
2:11
south korea medical device registration chapter 1 - overview
-
 1:50
1:50
conducting medical device clinical investigations in singapore
-
 53:33
53:33
a strategic roadmap assessing safety & biocompatibility of medical devices
-
 5:07
5:07
the regulatory framework for medical devices in other regions
-
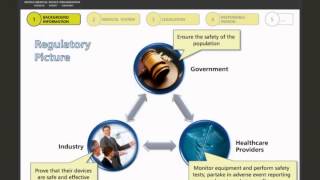 4:57
4:57
the hong kong regulatory system for medical devices
-
 53:15
53:15
medventions lecture series 2023: medical device & ai software regulation part 2