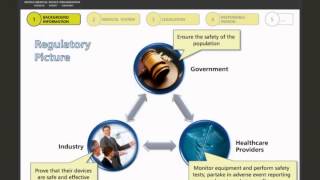
overview of the current medical device regulatory framework in singapore part 1 of 2
Published 11 years ago • 2.5K plays • Length 8:39Download video MP4
Download video MP3
Similar videos
-
 9:35
9:35
overview of the current medical device regulatory framework in singapore part 2 of 2
-
 2:58
2:58
overview of medical device registration
-
 8:39
8:39
class a medical devices regulatory framework enhancements part 1 of 4
-
 3:25
3:25
united states medical device registration chapter 3 - quality management system
-
 14:55
14:55
short course on the medical device regulation (eu) 2017/745
-
 23:18
23:18
risk basics for medical devices
-
 8:24
8:24
medical devices marketing: the power of medical devices marketing explained
-
 16:47
16:47
fda regulation of medical devices (part 1 of 3)
-
 4:57
4:57
the hong kong regulatory system for medical devices
-
 32:56
32:56
medical devices regulations webinar - 24 january 2023
-
 1:16:00
1:16:00
regulatory framework for medical devices & iv-vitro diagnosis part 1
-
 4:23
4:23
navigating the fda medical device classification process
-
 3:59
3:59
united states medical device registration chapter 1 - overview
-
 16:18
16:18
how is my medical device classified?
-
 13:30
13:30
medical device regulations and patient safety: comprehensive global policy.
-
 36:09
36:09
4.2 regulatory basics for medical devices
-
 1:50
1:50
conducting medical device clinical investigations in singapore
-
 3:50
3:50
medical device 101 - class i, ii, iii & de novo explained
-
 5:46
5:46
what is a medical device?
-
 3:27
3:27
e-labeling for medical device manufacturers - part 1
-
 2:11
2:11
south korea medical device registration chapter 1 - overview
-
 17:31
17:31
design control for medical devices - online introductory course