post-market surveillance for medical device industries in the eu: responsibilities and obligations
Published 3 years ago • 6.5K plays • Length 13:18Download video MP4
Download video MP3
Similar videos
-
 21:41
21:41
post-market surveillance as a medical device requirement in the eu
-
 47:15
47:15
understanding post-market surveillance requirements under eu mdr
-
 18:40
18:40
post market surveillance requirements under the new european medical device regulations
-
 1:37
1:37
purpose of the post-market surveillance process for medical devices
-
 2:59
2:59
what is post-market surveillance (pms) in the eu mdr?
-
 4:07
4:07
navigating the eu medical device regulation 2017/745
-
 1:25:47
1:25:47
eu postmarket surveillance requirements for medical devices
-
 1:24
1:24
post market surveillance for medical devices
-
 47:54
47:54
how to deal with the new post-market surveillance requirements under regulation (eu) 2017/745
-
 14:25
14:25
the importance of quality management systems in the medical device industry in the european union
-
 10:39
10:39
risk management in the medical device industry in the eu
-
 25:29
25:29
post-marketing surveillance
-
 13:56
13:56
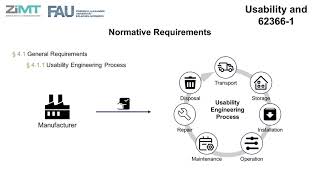
usability engineering in the medical device industry in the european union
-
 58:56
58:56
the new eu mdr pms requirements webinar
-
 45:13
45:13
webinar: how to set-up your post-market surveillance and vigilance system for ivds according to ivdr
-
 2:33
2:33
intelligent post-market surveillance for medical devices
-
 12:22
12:22
benefit-risk requirements in eu-mdr
-
 14:55
14:55
short course on the medical device regulation (eu) 2017/745
-
 57:13
57:13
us postmarket surveillance requirements for medical devices