risk management in the medical device industry in the eu
Published 2 years ago • 7.4K plays • Length 10:39Download video MP4
Download video MP3
Similar videos
-
 14:25
14:25
the importance of quality management systems in the medical device industry in the european union
-
 13:56
13:56
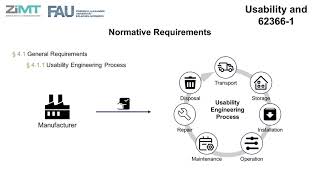
usability engineering in the medical device industry in the european union
-
 17:07
17:07
risk management for medical devices and iso 14971 - online introductory course
-
 13:18
13:18
post-market surveillance for medical device industries in the eu: responsibilities and obligations
-
 7:27
7:27
how to work with medical device risk management
-
 26:34
26:34
risk management in medical device development
-
 5:44
5:44
usability engineering and risk management for medical devices
-
 25:23
25:23
iso 14971: medical risk management best practices
-
 30:45
30:45
how to perform risk management for a software ?
-
 12:25
12:25
medical device software risk management and iec 62304 terminology
-
 0:33
0:33
risk management for medical device studies
-
 14:56
14:56
risk control measures in medical device software
-
 6:58
6:58
chemical risk management in eu - part 1
-
 54:08
54:08
getting to know changes of iso 14971 2019 risk management for medical devices
-
 51:00
51:00
regulatory standards & risk management in medical devices
-
 23:18
23:18
risk basics for medical devices
-
 25:01
25:01
what are the four different types of medical device risk analysis?
-
 15:22
15:22
how to estimate risk for a medical device according to iso 14971:2019
-
 3:34
3:34
risk management in medical devices
-
 10:25
10:25
iso 14971 & eu-mdr: residual risk requirements
-
 7:42
7:42
project risk management for medical devices: what’s to know?
-
 7:40
7:40
how to integrate proactive safety by design with medical device risk management