qms tip - update your listing of devices with the fda before october 1
Published 10 months ago • 185 plays • Length 0:58Download video MP4
Download video MP3
Similar videos
-
 0:47
0:47
qms tip - create a quality plan for implementing the fda qmsr
-
 22:34
22:34
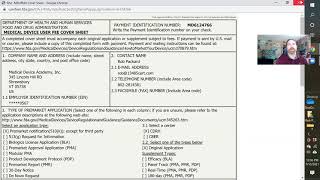
fda establishment registration and listing for medical devices
-
 0:43
0:43
qms tip - make your quality system product-specific.
-
 0:51
0:51
qms tip - make sure you look for changes in fda forms, not just changes to the regulations
-
 1:01
1:01
qms tip - when do you need a quality management system?
-
 21:07
21:07
fda renewal 2022
-
 3:38
3:38
medical device registration in malaysia
-
 3:21
3:21
qms software for medical devices | greenlight guru product overview
-
 3:25
3:25
united states medical device registration chapter 3 - quality management system
-
 15:04
15:04
cybersecurity documentation for a 510(k) submission
-
 0:58
0:58
qms tip - combine your ce marking procedure with your medical device file procedure
-
 0:54
0:54
qms tip - don't forget to confirm your subscription to our turnkey qms email list.
-
 0:39
0:39
qms tip - in your pms plan, ensure that you include risks.
-
 0:42
0:42
qms tip - include specific references to medwatch 3500a in your complaint handling form.
-
 0:41
0:41
qms tip - october is an ideal time to plan your audit schedule for next year.
-
 2:13
2:13
the fda's new medical device security requirements
-
 20:05
20:05
after successful fda approval, what do you need to do next?
-
 1:13:04
1:13:04
fda's proposed rule regarding device establishment registration & listing & how to register and list
-
 5:00
5:00
how to build a medical device qms using the best people, processes & technology (s.m.a.r.t system)
-
 0:15
0:15
this is the reality of becoming a surgeon.
-
 22:49
22:49
fda ecopy webinar