iso 13485: what you need to know to build a quality management systems for medical devices
Published 1 year ago • 5K plays • Length 13:11Download video MP4
Download video MP3
Similar videos
-
 3:14
3:14
what is a quality management system? - medical devices iso 13485 qms
-
 5:25
5:25
introduction to iso 13485 quality management system (qms) for medical devices
-
 13:27
13:27
quality management system vs quality assurance vs quality control: difference qa vs qc vs qms
-
 16:04
16:04
the 7 quality control (qc) tools explained with an example!
-
 12:35
12:35
overview of documented qms
-
 0:43
0:43
qms tip - make your quality system product-specific.
-
 11:59
11:59
your quick guide to iso 9001:2015 quality management system for beginner
-
 3:37
3:37
understanding quality management systems - what is iso 13485?
-
 14:25
14:25
the importance of quality management systems in the medical device industry in the european union
-
 0:15
0:15
qms tip - nonconformities
-
 0:47
0:47
qms tip - hazard identification shortcut for medical devices...
-
 9:05
9:05
what is quality management system (qms) | elements of quality management system
-
 0:50
0:50
qms tip - what does qms mean?
-
 2:08
2:08
what is qms | quality management system explained
-
 12:57
12:57
medical device quality management system md qms(iso13485:2016)
-
 7:52
7:52
what is a quality management system (qms)?
-
 0:51
0:51
sys-024 capa procedure training (july 22)
-
 45:27
45:27
qms from a regulatory perspective
-
 0:58
0:58
qms tip - update your listing of devices with the fda before october 1
-
 0:14
0:14
qms tip - virtual qms vs eqms?
-
 14:22
14:22
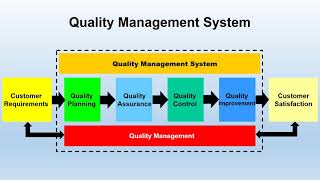
what is quality management system (qms)?