rate law of a reaction is experimentally determined as rate = k [a]^2 [b]^0 [c]the unit of k is ___…
Published 5 months ago • 1 view plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 34:53
34:53
chemical kinetics - initial rates method
-
 9:10
9:10
kinetics: initial rates and integrated rate laws
-
 18:48
18:48
writing rate laws of reaction mechanisms using the rate determining step - chemical kinetics
-
 48:46
48:46
integrated rate laws - zero, first, & second order reactions - chemical kinetics
-
 10:49
10:49
solving a rate law using the initial rates method
-
 3:42
3:42
how to find the rate law and rate constant (k)
-
 31:50
31:50
collision theory - arrhenius equation & activation energy - chemical kinetics
-
 6:53
6:53
the rate constant
-
 4:51
4:51
reaction rate law (example)
-
 9:29
9:29
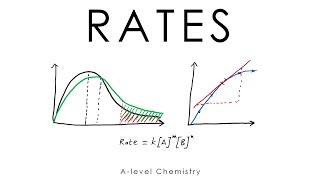
rate of reaction - a-level chemistry
-
 5:35
5:35
chemistry - chemical kinetics (2 of 30) reaction rate- definition
-
 4:28
4:28
elementary rate laws - unimolecular, bimolecular and termolecular reactions - chemical kinetics
-
 18:03
18:03
how to solve non-elementary reaction rate problems
-
 0:33
0:33
1. the rate law of the reaction no2 with co to produce no and co2 is second order with respect to n…
-
 4:29
4:29
how to solve reaction rate problems
-
 8:44
8:44
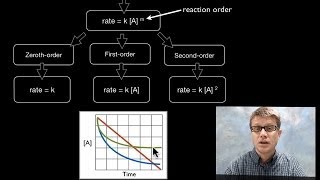
the rate law
-
 0:33
0:33
the rate constant k is dependent on i. the concentration of the reactant ii: the nature of the reac…
-
 5:35
5:35
elementary reactions | kinetics | ap chemistry | khan academy
-
 4:03
4:03
determining rate order
-
 0:33
0:33
consider the reaction: 2 hgcl2 (aq) c2o4^2-(aq) - 2 cl^-(aq) 2 co2 (g) hg2cl2(s). the initia…
-
 13:19
13:19
rate law
-
 0:33
0:33
a reaction in which a, b, and c react to form products is first order in a, second order in b, and …