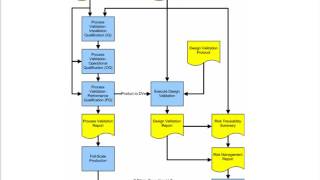
risk-based verification and validation planning to meet us fda and iso 13485 requirements
Published 2 years ago • 169 plays • Length 15:43Download video MP4
Download video MP3
Similar videos
-
 8:56
8:56
process validation or verification for your medical device (iso 13485)
-
 36:04
36:04
how to validate an eqms for medical devices? (iso 13485 - fda qsr)
-
 32:09
32:09
fda aligns qmsr with iso 13485?
-
 17:31
17:31
design control for medical devices - online introductory course
-
 10:39
10:39
risk management in the medical device industry in the eu
-
 15:22
15:22
how to estimate risk for a medical device according to iso 14971:2019
-
 1:28:00
1:28:00
process validation for medical device manufacturers
-
 1:02:02
1:02:02
verification & testing strategies for compliance with iso 13485:2016, iec 62304 / 60601-1 / 82304-1
-
 4:30
4:30
sys-044 software development and validation procedure
-
 3:24
3:24
software validation 820.30g & iso 13485 § 4.1.6 & 7.3.7 (executive series #20)
-
 49:52
49:52
qmsr, iso 13485, & risk management
-
 59:28
59:28
2021-06-16 verification and validation by geoff sizer
-
 12:49
12:49
process validation for medical devices - short course
-
 1:52:50
1:52:50
md-qms full course of iso 13485:2016 | training on iso 13485:2016| training on full course |
-
 23:18
23:18
risk basics for medical devices
-
 1:25:43
1:25:43
how to simplify your compliance with the new iso 13485:2016
-
 25:45
25:45
how to get iso 13485 certified? (quality management system)
-
 4:25
4:25
medical devices: how to verify iso 13485 certificates?
-
 57:11
57:11
webinar // cybersecurity in the medical devices – how to show compliance to regulatory requirements