the 510k process - straight talk with susan: key issues in medical technology regulation
Published 4 years ago • 178 plays • Length 1:24Download video MP4
Download video MP3
Similar videos
-
 1:43
1:43
510(k) clinical testing - straight talk with susan: key issues in medical technology regulation
-
 2:13
2:13
fda's mission - straight talk with susan: key issues in medical technology regulation
-
 3:01
3:01
industry-fda interactions - straight talk with susan: key issues in medical technology regulation
-
 2:10
2:10
patient information - straight talk with susan: key issues in medical technology regulation
-
 1:35
1:35
risk and medical technology - straight talk with susan: key issues in medical technology regulation
-
 1:43
1:43
clinical trials and medtech - straight talk with susan: key issues in medical technology regulation
-
 2:44
2:44
regulation of medtech vs drugs - straight talk with susan: key issues in medtech regulation
-
 1:19:59
1:19:59
the 510(k) submission: requirements, contents, and options
-
 13:35
13:35
combimax - a complete energy workstation
-
 4:34
4:34
dexis™ is scanflow - using the occlusion check & undercut check tools
-
 2:16
2:16
concerns about 510(k) process
-
 2:17
2:17
basics of 510(k) clearance process
-
 2:19
2:19
understanding the fda medical device 510k process
-
 32:06
32:06
the fda 510k program
-
 11:06
11:06
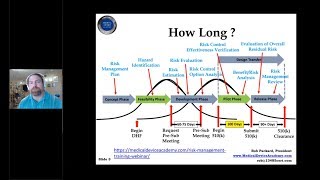
how to prepare a medical device 510k submission for fda
-
 1:34:30
1:34:30
how to prepare a medical device 510k submission for fda | rob packard | joe hage
-
 17:20
17:20
fda estar - general info - #1 in the 510(k) estar series
-
 1:06:28
1:06:28
mastering your 510(k) submission process
-
 1:19:59
1:19:59
requirements contents and options : the 510k submission
-
 16:57
16:57
what is the status of the 510(k) course?
-
 6:34
6:34
510(k) submission predicate selection webinar - updated for 2022