the relation between molarity (m) and molality (m) is given by: (ρ=. density of solution (g / ml)...
Published 2 years ago • 144 plays • Length 4:58Download video MP4
Download video MP3
Similar videos
-
 6:01
6:01
the relation between molarity (m) and molality (m) is given by : ( ρ= density of solution (g / ml...
-
 4:40
4:40
the relation between molarity (m) and molality (m) is given by : (p=density of solution (g/ml), ...
-
 2:59
2:59
the relation between molarity (m) and molality (m) is given by : (p=density of solution (g/ml), ...
-
 3:00
3:00
the relation between molarity (m) and molality (m) is given by : (p=density of solution (g
-
 25:23
25:23
colligative properties - boiling point elevation, freezing point depression & osmotic pressure
-
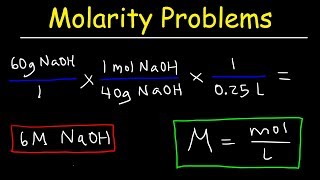 21:27
21:27
molarity practice problems
-
 9:36
9:36
trick to calculate molarity | molarity practice problems
-
 6:01
6:01
the relation between molarity \( (m) \) and molality \( (m) \) is g...
-
 6:01
6:01
the relation between molarity \( (m) \) and molality \( (m) \) is g...
-
 6:01
6:01
the relation between molarity \( (m) \) and molality \( (m) \) is g...
-
 34:54
34:54
solution in chemistry lecture 02 | chemistry class 12 | neet 2025 | lecturer polytechnic lecturer
-
 6:01
6:01
the relation between molarity \( (m) \) and molality \( (m) \) is g...
-
 31:25
31:25
molarity, molality, volume & mass percent, mole fraction & density - solution concentration problems
-
 6:01
6:01
the relation between molarity \( (m) \) and molality \( (m) \) is g...
-
 6:07
6:07
the correct relationship between molarity (m) and molality (m) is ( d = density of the solution,...
-
 4:38
4:38
the relation between molarity (c ) and molality (m) is given by )d= density of solution, m =mola...
-
 4:39
4:39
calculate molality given density and percent by weight/mass
-
 0:57
0:57
a substance having equal number of molecules as in 9gm of water is? aiims vs iit #shorts #neet #jee
-
 9:54
9:54
relation between molarity and density of solution | #molarityimpulseiitjee
-
 8:23
8:23
the correct relationship between molarity and molality is.
-
 0:15
0:15
part-4 solution short notes ( relation between density, molarity and molality and related question)
-
 4:07
4:07
difference between molarity and molality