what’s new in the draft fda cybersecurity guidance?
Published Streamed 2 years ago • 935 plays • Length 27:49Download video MP4
Download video MP3
Similar videos
-
 46:31
46:31
fda cybersecurity testing requirements - interview with red sentry
-
 1:08:44
1:08:44
cybersecurity webinar - learn what the fda wants in your 510(k)
-
 4:07
4:07
fda’s new guidance on cybersecurity for medical devices
-
 7:32
7:32
fda finally released the 2023 cybersecurity final guidance document
-
 22:27
22:27
medical device cybersecurity: understanding the fda guidelines with mike kijewski from medcrypt
-
 16:50
16:50
important new fda guidance – coming soon
-
 37:47
37:47
using the new estar templates for a 510(k) submission and the fda estar draft guidance
-
 22:17
22:17
dfs101: 2.1 cybersecurity
-
 59:31
59:31
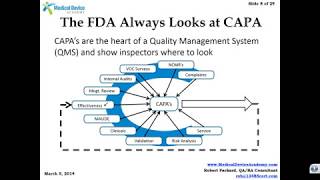
how to prepare for your next fda inspection
-
 1:02:57
1:02:57
fda quality systems regulation requirements - regulatory documents explained
-
 18:25
18:25
what do you need for cybersecurity risk management?
-
 5:25
5:25
cloud and medical device cybersecurity: the fda’s 2023 guidelines
-
 9:23
9:23
fda's medical device cybersecurity guidance
-
 50:36
50:36
fda cybersecurity in medical devices
-
 38:05
38:05
new fda requirements for connected medical devices. how cybersecurity impacts regulatory approval
-
 1:28:37
1:28:37
fda cybersecurity and software policy updates: navigating the new fda guidance documents for medtech
-
 12:18
12:18
where to find fda software and cybersecurity submission requirements
-
 15:04
15:04
cybersecurity documentation for a 510(k) submission
-
 15:37
15:37
what’s in attachment g of the new fda biocompatibility guidance?
-
 43:09
43:09
usenix security '18 - medical device cybersecurity through the fda lens
-
 11:57
11:57
medical device cybersecurity regulatory update