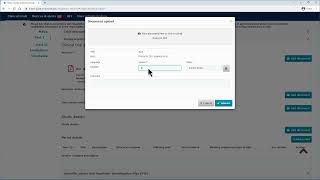
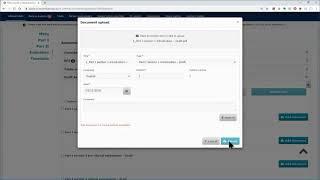
ctis – m10 how to submit an initial cta in the ctis sponsor workspace – fill in the part i section
Published 3 years ago • 11K plays • Length 10:03Download video MP4
Download video MP3
Similar videos
-
 4:14
4:14
ctis – m10 how to submit an initial cta in the ctis – fill in the trial details of part i section.
-
 4:00
4:00
ctis – m10 how to submit an initial cta in the ctis – fill in the sponsor details of part i section.
-
 3:35
3:35
ctis – m10 how to submit an initial cta in the ctis – fill in the product details of part i section.
-
 4:52
4:52
ctis – m10 how to submit an initial cta in the ctis – fill in the form and the msc sections.
-
 5:20
5:20
ctis – m10 how to submit an initial cta in the ctis – fill in the part ii section
-
 3:41
3:41
ctis – m10 how to submit an additional msc application in the ctis sponsor workspace
-
 7:43
7:43
ctis – m10 how to submit a single trial substantial modification in the ctis sponsor workspace
-
 6:39
6:39
ctis – m08 how to assess an initial clinical trial application in ctis – part i
-
 5:15
5:15
stelmec!!new macnisum install(install)
-
 12:43
12:43
selpro :pt16 how to program hmi in selec plc
-
 3:28
3:28
product of the week-tilite
-
 16:00
16:00
implementation of the clinical trials regulation
-
 5:30
5:30
ctis - m15 how to view and download cts in the authority workspace
-
 3:25
3:25
ctis - m06 how to access an initial cta in ctis – validation - rfi and issue validation decision
-
 1:00:14
1:00:14
preparing content for ctis & the eu ctr
-
 5:58
5:58
ctis – m07 how to request roles and how to assign roles to registered users in ctis
-
 59:36
59:36
beyond first impressions: ctis lessons learned in first 3 months
-
 3:30
3:30
ctis - m02 ctis specific functionalities
-
 1:28:56
1:28:56
ctis bitesize talk: transitional trials and additional msc application
-
 3:22
3:22
ctis - m03 registration of a new organisation in ctis
-
 4:43
4:43
ctis – m08 how to assess an initial clinical trial application in ctis – part ii