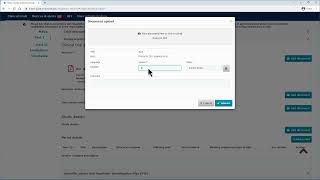
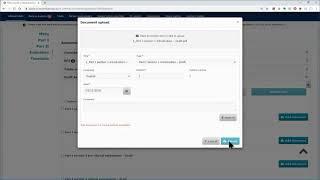
ctis – m10 how to submit an initial cta in the ctis – fill in the part ii section
Published 3 years ago • 10K plays • Length 5:20Download video MP4
Download video MP3
Similar videos
-
 4:52
4:52
ctis – m10 how to submit an initial cta in the ctis – fill in the form and the msc sections.
-
 10:03
10:03
ctis – m10 how to submit an initial cta in the ctis sponsor workspace – fill in the part i section
-
 4:14
4:14
ctis – m10 how to submit an initial cta in the ctis – fill in the trial details of part i section.
-
 3:35
3:35
ctis – m10 how to submit an initial cta in the ctis – fill in the product details of part i section.
-
 4:00
4:00
ctis – m10 how to submit an initial cta in the ctis – fill in the sponsor details of part i section.
-
 3:41
3:41
ctis – m10 how to submit an additional msc application in the ctis sponsor workspace
-
 7:43
7:43
ctis – m10 how to submit a single trial substantial modification in the ctis sponsor workspace
-
 4:43
4:43
ctis – m08 how to assess an initial clinical trial application in ctis – part ii
-
 4:16
4:16
cheque clearing process - cts in india by elogic technologies
-
 3:34
3:34
hướng dẫn hiệu chuẩn đầu cân cti-1110
-
 2:59
2:59
how to set the ct rate on the eastron sdm630mct ml series
-
 6:39
6:39
ctis – m08 how to assess an initial clinical trial application in ctis – part i
-
 3:25
3:25
ctis - m06 how to access an initial cta in ctis – validation - rfi and issue validation decision
-
 1:27:04
1:27:04
clinical trials information system ctis bitesize talk requests for information
-
 2:01
2:01
clinical trials in the eu
-
 0:31
0:31
clinical trials in the eu - short version
-
 3:12:21
3:12:21
sme and academia 2 part training webinar: day 2
-
 45:08
45:08
clinical trials information system ctis walk in clinic
-
 45:53
45:53
clinical trials information system ctis: walk in clinic
-
 4:56
4:56
ctis - m17 how to create an ad hoc assessment – discussion with mss and create rfis
-
 1:27:14
1:27:14
clinical trials information system (ctis) bitesize talk: notifications - part 1
-
 27:45
27:45
interview: impact of ctis on tmf management