do you know how to avoid receiving an fda rta hold letter?
Published 3 years ago • 152 plays • Length 3:28Download video MP4
Download video MP3
Similar videos
-
 22:03
22:03
learn how to avoid receiving an rta hold letter when you submit your 510(k) submission to the fda?
-
 1:12:32
1:12:32
rta hold and ai request responses to an fda 510(k) reviewer
-
 3:27
3:27
how to respond to an fda rta hold - new blog posting
-
 0:17
0:17
what do you want to know about the fda 510(k) process?
-
 21:28
21:28
how do you write a regulatory pathway analysis?
-
 21:42
21:42
how to respond to a an fda additional information (ai) request
-
 0:16
0:16
three 510(k) tips
-
 49:08
49:08
medical devices 101: an entry level overview of the fda
-
 9:17
9:17
analytical method development in pharmaceutical industry l 21 basic and important interview question
-
 7:13
7:13
what is 21 cfr 820?
-
 51:46
51:46
510(k) frequently asked questions
-
 35:16
35:16
fda udi regulation’s impact on medical device labelers webinar
-
 38:26
38:26
how to prepare an fda estar 510(k) submission
-
 47:11
47:11
fda refused 510(k) submissions
-
 0:53
0:53
510(k) tip - make sure you are signed up for email notifications of new fda guidance documents
-
 22:49
22:49
fda ecopy webinar
-
 52:43
52:43
webinar for special 510(k) submissions
-
 8:51
8:51
human factors and usability testing for a 510(k) submission
-
 37:47
37:47
using the new estar templates for a 510(k) submission and the fda estar draft guidance
-
 4:11
4:11
how long does a 510(k) actually take?
-
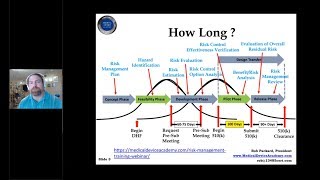 1:34:30
1:34:30
how to prepare a medical device 510k submission for fda | rob packard | joe hage