how to prepare a 510(k) quality submission
Published 8 years ago • 1.2K plays • Length 1:07:58Download video MP4
Download video MP3
Similar videos
-
 11:06
11:06
how to prepare a medical device 510k submission for fda
-
 1:19:59
1:19:59
requirements contents and options : the 510k submission
-
 1:34:30
1:34:30
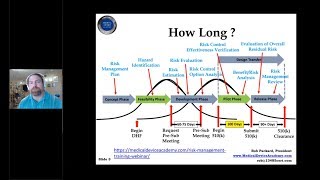
how to prepare a medical device 510k submission for fda | rob packard | joe hage
-
 1:19:59
1:19:59
the 510(k) submission: requirements, contents, and options
-
 12:48
12:48
preparing an fda 510 (k) submission often called 510k
-
 6:34
6:34
510(k) submission predicate selection webinar - updated for 2022
-
 1:34:30
1:34:30
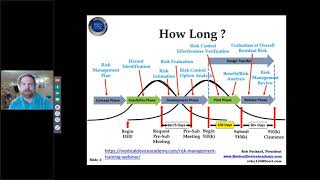
how to prepare a medical device 510k submission for fda | rob packard | joe hage | updated
-
 38:26
38:26
how to prepare an fda estar 510(k) submission
-
 57:47
57:47
fda 101 for medical devices
-
![[2022년 해외 수출국 최신 규제 전문가 세미나] 02. 미국 인증 fda(510k) 인허가 대응방안](https://i.ytimg.com/vi/F9HFv6Y0mOQ/mqdefault.jpg) 1:59:24
1:59:24
[2022년 해외 수출국 최신 규제 전문가 세미나] 02. 미국 인증 fda(510k) 인허가 대응방안
-
 49:08
49:08
medical devices 101: an entry level overview of the fda
-
 2:19
2:19
how to prepare and submit a bullet proof 510(k) submission
-
 10:05
10:05
bullet proof 510(k) - latest fda proposed changes to the process - globalcompliancepanel
-
 37:47
37:47
using the new estar templates for a 510(k) submission and the fda estar draft guidance
-
 1:06:28
1:06:28
mastering your 510(k) submission process
-
 1:36:58
1:36:58
how to prepare a 510k submission for fda
-
 52:43
52:43
webinar for special 510(k) submissions
-
 15:04
15:04
cybersecurity documentation for a 510(k) submission
-
 0:16
0:16
510(k) tip - saving your work
-
 40:11
40:11
510k workshop: how to prepare an fda submission (part i)
-
 47:11
47:11
fda refused 510(k) submissions
-
 4:11
4:11
what is the difference between a 510k and de novo?